Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Dalchem | Russia | |||
|---|---|---|---|---|
 | www.dalchem.com | |||
 | +7 (8312) 753-772 | |||
 | +7 (8312) 750-799 | |||
 | dregichy@dalchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Takasago International Corp. (USA) | USA | |||
|---|---|---|---|---|
 | www.takasago.com | |||
 | +1 (201) 767-9001 | |||
 | +1 (201) 784-7277 | |||
 | aromachemicals@takasago.com | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Phosphine ligand |
|---|---|
| Name | (S)-(-)-2,2'-Bis(di-p-tolylphosphino)-1,1'-binaphthyl |
| Synonyms | [1-[2-bis(4-methylphenyl)phosphanylnaphthalen-1-yl]naphthalen-2-yl]-bis(4-methylphenyl)phosphane |
| Molecular Structure | 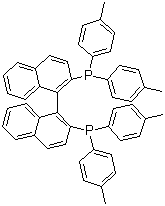 |
| Molecular Formula | C48H40P2 |
| Molecular Weight | 678.78 |
| CAS Registry Number | 99646-28-3 |
| EC Number | 677-521-0 |
| SMILES | CC1=CC=C(C=C1)P(C2=CC=C(C=C2)C)C3=C(C4=CC=CC=C4C=C3)C5=C(C=CC6=CC=CC=C65)P(C7=CC=C(C=C7)C)C8=CC=C(C=C8)C |
| Melting point | 255-257 °C |
|---|---|
| alpha | 156 |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details |
| SDS | Available |
|
(S)-(-)-2,2'-Bis(di-p-tolylphosphino)-1,1'-binaphthyl is a chiral ligand renowned for its pivotal role in asymmetric catalysis. This compound features a binaphthyl backbone, which provides a rigid, chiral environment for its phosphine groups. The ligand's structure is specifically designed to enhance enantioselectivity in catalytic processes, making it highly valuable in both academic research and industrial applications. The discovery of (S)-(-)-2,2'-Bis(di-p-tolylphosphino)-1,1'-binaphthyl was driven by the need for more efficient chiral ligands that could provide high levels of enantioselectivity in various asymmetric reactions. The synthesis of this ligand involves the coupling of (S)-(-)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl with p-tolyl groups. This modification enhances the ligand's steric and electronic properties, leading to improved catalytic performance. One of the most significant applications of (S)-(-)-2,2'-Bis(di-p-tolylphosphino)-1,1'-binaphthyl is in asymmetric hydrogenation. The ligand's ability to create a chiral environment around the metal center allows for the efficient reduction of prochiral substrates to yield enantiomerically pure products. This application is particularly valuable in the pharmaceutical industry, where the synthesis of chiral drugs is critical. Beyond hydrogenation, (S)-(-)-2,2'-Bis(di-p-tolylphosphino)-1,1'-binaphthyl is also used in other asymmetric transformations, including hydroformylation and olefin metathesis. Its versatility in these reactions demonstrates its broad applicability in organic synthesis. The ligand's effectiveness in these processes is attributed to its ability to stabilize metal centers and promote selective interactions with substrates. The success of (S)-(-)-2,2'-Bis(di-p-tolylphosphino)-1,1'-binaphthyl in various catalytic processes highlights its importance in the field of asymmetric catalysis. Researchers continue to explore its potential in new catalytic systems and reactions, aiming to further enhance its performance and expand its applications. The development of this ligand represents a significant advancement in the design of chiral ligands and their use in fine chemical synthesis. In conclusion, (S)-(-)-2,2'-Bis(di-p-tolylphosphino)-1,1'-binaphthyl is a key chiral ligand that has greatly impacted asymmetric catalysis. Its ability to provide high enantioselectivity in hydrogenation and other asymmetric reactions underscores its value in both research and industrial applications. The ongoing exploration of its capabilities continues to contribute to advancements in the field of organic synthesis. References 2003. Mechanism of Asymmetric Hydrogenation of Ketones Catalyzed by BINAP/1,2-Diamine−Ruthenium(II) Complexes. Journal of the American Chemical Society, 125(41). DOI: 10.1021/ja030272c 2005. Asymmetric Hydrogenation of tert-Alkyl Ketones. Journal of the American Chemical Society, 127(20). DOI: 10.1021/ja052071+ 2015. 1,2- Versus 1,4-Asymmetric Reduction of Ketones. Progress in Enantioselective Cu(I)-catalyzed Formation of Stereogenic Centers. DOI: 10.1007/3418_2015_157 |
| Market Analysis Reports |