Online Database of Chemicals from Around the World
| Shanghai Oripharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oripharm.com.cn | |||
 | +86 (21) 6439-6936 | |||
 | +86 (21) 6481-4386 | |||
 | info@oripharm.com.cn sales@oripharm.com.cn | |||
| Chemical distributor since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Nanjing Search Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.searchbio.com.cn | |||
 | +86 (25) 8168-2922 8586-0978 +86 18913919581 | |||
 | +86 (25) 8586-0962 | |||
 | trade@searchbio.com.cn sales@searchbio.com.cn Linda@searchbio.com.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Beijing TND Science and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tndtech.com.cn | |||
 | +86 (10) 8075-1706 +86 13521671946 | |||
 | +86 (10) 8075-1706 | |||
 | sales@tndtech.com.cn | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Hubei Jianchu Biomedical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hbhxry.com | |||
 | +86 (027) 6538-1247 +86 15671591110 | |||
 | +86 (027) 6552-6669 | |||
 | 2394105454@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2022 | ||||
| J&H Chemical HK Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jhechem.com | |||
 | +86 (571) 8739-6430 8739-6432 | |||
 | +86 (571) 8739-6431 | |||
 | sales@jhechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Dalo Chem Life Sciences (Dongguan) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dalochem.com | |||
 | +86 15019076757 | |||
 | sales@dalochem.com | |||
 | WhatsApp:+86-15019076757 | |||
| Chemical distributor since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| AlzChem Trostberg GmbH | Germany | |||
|---|---|---|---|---|
 | www.alzchem.com | |||
 | +49 (8) 621-860 | |||
 | +49 (8) 621-2911 | |||
 | brigitte.weiher@alzchem.com | |||
| Chemical manufacturer since 1908 | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Classification | Chemical reagent >> Organic reagent >> Guanidine salt |
|---|---|
| Name | Guanidine thiocyanate |
| Synonyms | Guanidinium thiocyanate |
| Molecular Structure | 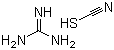 |
| Molecular Formula | C2H6N4S;CH5N3.HSCN |
| Molecular Weight | 118.16 |
| CAS Registry Number | 593-84-0 |
| EC Number | 209-812-1 |
| SMILES | C(#N)S.C(=N)(N)N |
| Density | 1.29 g/mL |
|---|---|
| Melting point | 118-122 °C |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312+H332-H302-H312-H314-H318-H332-H412 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P330-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Guanidine thiocyanate (GITC) is a powerful chaotropic agent that was first synthesized in the mid-20th century. It was developed while exploring the unique properties of guanidine derivatives to disrupt molecular structure. GITC quickly gained fame for its powerful ability to denature proteins and nucleic acids and is of great value in molecular biology and biochemistry. The chemical formula of guanidine thiocyanate is CH5N3•CHNS. It is a colorless to pale yellow crystalline solid that is highly soluble in water and organic solvents. The compound has a guanidine group attached to a thiocyanate ion, which gives it its powerful chaotropic properties. This structure disrupts hydrogen bonds, effectively denaturing biomolecules. GITC exerts its chaotropic effect by disrupting the hydrogen-bonding network of water, disrupting the hydrophobic interactions that maintain the native structure of proteins and nucleic acids. This disruption results in unfolding of proteins and denaturation of nucleic acids, thereby facilitating their study and manipulation in various biochemical applications. GITC is widely used in RNA isolation protocols. It denatures RNases, protecting RNA from degradation. This makes GITC essential for extracting high-quality RNA from biological samples, critical for applications such as reverse transcription, quantitative PCR (qPCR), and RNA sequencing. GITC is used to denature proteins, facilitating studies of protein folding, stability, and interactions. By disrupting secondary and tertiary structure, researchers can analyze primary structure and identify protein domains and functional sites. GITC is used in DNA purification to remove proteins and other contaminants. Its ability to denature proteins ensures that DNA samples are free of enzymatic activity and other impurities, which is critical for downstream applications such as cloning, sequencing, and PCR. GITC is used to inactivate viruses in clinical samples, ensuring the safety of laboratory personnel and preserving nucleic acids for diagnostic testing. During the COVID-19 pandemic, this application is particularly important for preparing samples for SARS-CoV-2 testing. Guanidine thiocyanate is a hazardous substance and must be handled with care. It is irritating to the skin, eyes, and respiratory tract and can be toxic if ingested or inhaled. Appropriate personal protective equipment, such as gloves, lab coats, and goggles, should be worn when handling GITC. Additionally, solutions should be prepared in a well-ventilated area or under a fume hood to avoid inhalation of vapors. References 2024. Destabilization of the D2 domain of Thermotoga maritima arginine binding protein induced by guanidinium thiocyanate and its counteraction by stabilizing agents. Protein science : a publication of the Protein Society, 33(9). DOI: 10.1002/pro.5146 2023. Back to Old Books Toward Affordable Research: Homemade Phenol-Based Reagent for Triphasic RNA Purification. Biochemical Genetics, 62(3). DOI: 10.1007/s10528-023-10509-3 2022. RNA Extraction from Plant Tissue with Homemade Acid Guanidinium Thiocyanate Phenol Chloroform (AGPC). Current Protocols, 2(1). DOI: 10.1002/cpz1.351 |
| Market Analysis Reports |