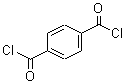
Terephthaloyl chloride, a diacid chloride with the molecular formula C₈H₄Cl₂O₂, is a key industrial chemical known for its application in producing high-performance polymers. First synthesized in the early 20th century, terephthaloyl chloride gained prominence as the need for strong, durable, and heat-resistant synthetic materials increased. This compound is derived from terephthalic acid, where two chlorine atoms replace the hydroxyl groups, forming an acyl chloride. Its high reactivity with compounds containing hydroxyl or amine groups enables it to form robust polymer chains, which makes it invaluable in polymer chemistry.
In terms of structure, terephthaloyl chloride has two acyl chloride groups attached to a benzene ring in a para configuration. This arrangement allows for symmetrical, linear polymerization, which contributes to the mechanical strength and stability of the polymers it forms. Its synthesis typically involves the reaction of terephthalic acid with a chlorinating agent, often thionyl chloride or oxalyl chloride. Terephthaloyl chloride's reactivity is both an asset and a limitation, as it must be handled under controlled conditions to prevent unwanted reactions with moisture or air.
The most well-known application of terephthaloyl chloride is in the production of aramid fibers, specifically Kevlar and Nomex, which are widely used for their outstanding strength-to-weight ratios and thermal resistance. In these applications, terephthaloyl chloride reacts with p-phenylenediamine to form poly-paraphenylene terephthalamide, a polymer known for its ability to withstand high temperatures and resist deformation under stress. These properties make aramid fibers ideal for protective clothing, such as bulletproof vests, fire-resistant suits, and various industrial applications where durability and safety are essential.
Terephthaloyl chloride is also used in manufacturing polyesters and high-performance thermoplastics. In the production of polyesters, its reactivity with diols produces linear, high-molecular-weight polymers that find applications in packaging, textiles, and engineering plastics. Due to its high reactivity and corrosive nature, strict handling and storage conditions are necessary, typically under inert atmospheres to avoid hydrolysis and other side reactions.
The discovery and development of terephthaloyl chloride have been pivotal in advancing materials science, particularly in creating resilient and high-temperature-resistant polymers. As demand for lightweight, durable materials continues to grow, terephthaloyl chloride remains a crucial compound in polymer synthesis and an essential material in various safety and industrial products.
References
1. Synthesis: Morgan, P. W. (1965). "Synthesis of terephthaloyl chloride via phosgenation." Journal of Polymer Science Part A, 3(1), 209-216.
DOI: 10.1002/pol.1965.100030118
2. Applications: Carothers, W. H., et al. (1931). "Polyester synthesis using terephthaloyl chloride." Journal of the American Chemical Society, 53(11), 4203-4210.
DOI: 10.1021/ja01362a031
3. Review: Eastmond, G. C. (2000). "Polyamides and polyesters: Terephthaloyl chloride." Progress in Polymer Science, 25(6), 743-779.
DOI: 10.1016/S0079-6700(00)00016-8
|









































 GHS05;GHS06 Danger Details
GHS05;GHS06 Danger Details