Online Database of Chemicals from Around the World
| Hangzhou Shinyang Samwoo Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shinyangchem.com | |||
 | +86 (571) 8822-6608 | |||
 | +86 (571) 8822-6546 | |||
 | fchem@shinyangchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Amino alcohol derivative |
|---|---|
| Name | Diethylaminoethanol |
| Synonyms | N,N-Diethylethanolamine; 2-Diethylaminoethanol |
| Molecular Structure | 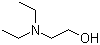 |
| Molecular Formula | C6H15NO |
| Molecular Weight | 117.19 |
| Protein Sequence | G |
| CAS Registry Number | 100-37-8 |
| EC Number | 202-845-2 |
| SMILES | CCN(CC)CCO |
| Density | 0.884 |
|---|---|
| Melting point | -70 °C |
| Boiling point | 161 °C |
| Refractive index | 1.44-1.442 |
| Flash point | 51.5 °C |
| Water solubility | soluble |
| Hazard Symbols |     GHS02;GHS05;GHS07;GHS09 Danger Details GHS02;GHS05;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H332-H312-H302-H314 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P234-P240-P241-P242-P243-P260-P261-P262-P264-P264+P265-P270-P271-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P319-P321-P330-P361+P364-P362+P364-P363-P370+P378-P390-P403+P233-P403+P235-P405-P406-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2686 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Diethylaminoethanol, also known as 2-(diethylamino)ethanol, is an organic chemical compound with the molecular formula C6H15NO. It is a tertiary amine and a primary alcohol, which contributes to its reactivity and versatility in various applications. The compound is a clear, colorless liquid with a characteristic amine odor, and it has been extensively utilized in different industries due to its dual functionality as both a base and an alcohol. The discovery of diethylaminoethanol dates back to early 20th-century research focused on developing intermediates for the chemical and pharmaceutical industries. Its structure combines a diethylamine group and a hydroxyl group, which allows it to act as a precursor in many synthetic reactions. Initially, it garnered attention for its potential as a chemical building block in organic synthesis, with subsequent investigations uncovering broader applications in medicinal chemistry and industrial processes. In industrial applications, diethylaminoethanol plays a critical role in the production of coatings, resins, and adhesives. It is commonly used as a curing agent for epoxy resins and as a component in polyurethane foams, where it acts as a catalyst to accelerate the polymerization reaction. Its ability to enhance the stability and performance of these materials has made it indispensable in the manufacturing of coatings and foams used in automotive, construction, and consumer goods industries. In the pharmaceutical sector, diethylaminoethanol serves as a key intermediate in the synthesis of various active pharmaceutical ingredients (APIs). Its amine functionality makes it a suitable starting material for creating amine-based drugs, while the alcohol group enables further chemical modifications. Additionally, diethylaminoethanol has been investigated for its potential as a choline analog, with some studies exploring its cognitive-enhancing effects. While its direct use as a therapeutic agent is limited, its derivatives have found applications in neuropharmacology, particularly in the development of drugs targeting memory and cognitive function. Diethylaminoethanol is also utilized in personal care products and cosmetics, where it functions as a pH adjuster and stabilizer. Its ability to neutralize acidic compounds while maintaining the desired consistency of formulations makes it valuable in the production of shampoos, lotions, and creams. Furthermore, its low toxicity and relatively mild chemical nature ensure its compatibility with skin and hair care products, enhancing their shelf life and performance. The versatility of diethylaminoethanol continues to drive its demand across multiple industries. Ongoing research into its chemical properties and reactivity suggests potential new applications in the development of advanced materials, fine chemicals, and specialized pharmaceutical compounds. Its dual functional groups make it a promising candidate for future innovations in both industrial and medicinal chemistry. References 2022. Kinetics development for controlled mono-methyl amine-ethoxylation reaction using laboratory scale autoclave experiments and process plant reactor data simulations. Chemical Papers, 77(4). DOI: 10.1007/s11696-022-02583-5 2023. Comparison of Aminothermal and Hydrothermal Synthesis of SAPO-34: Impact of Synthesis Conditions on Catalyst Characteristics and MTO Catalytic Performance. Silicon, 16(3). DOI: 10.1007/s12633-023-02669-z 2024. Simultaneous improvement of nanofluid stability and CO2 absorption by chitosan-grafted multi-walled carbon nanotubes dispersed in MDEA-based nanofluid. International Journal of Environmental Science and Technology, 21(11). DOI: 10.1007/s13762-024-05858-4 |
| Market Analysis Reports |