Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Amino alcohol derivative |
|---|---|
| Name | 2-(Diethylamino)ethanol hydrochloride |
| Synonyms | Dehydasal; Perdilaton |
| Molecular Structure | 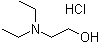 |
| Molecular Formula | C6H15NO.HCl |
| Molecular Weight | 153.65 |
| Protein Sequence | C |
| CAS Registry Number | 14426-20-1 |
| EC Number | 238-395-9 |
| SMILES | CCN(CC)CCO.Cl |
| Melting point | 135-136 °C* |
|---|---|
| * | Blicke, F. F. |
| SDS | Available |
|---|---|
|
2-(Diethylamino)ethanol hydrochloride, commonly known as diethylaminoethanol hydrochloride, is a synthetic compound that belongs to the class of amino alcohols. It is characterized by the presence of a diethylamino group and an ethanol moiety, which contributes to its unique properties and applications. The discovery of 2-(diethylamino)ethanol hydrochloride can be traced back to the early 20th century when researchers were exploring various amino alcohols for their potential therapeutic uses. Its hydrochloride form was later synthesized to enhance its solubility and stability in aqueous solutions, making it more suitable for pharmaceutical applications. The compound has gained attention due to its biological activity and has been investigated for its effects on the central nervous system. One of the primary applications of 2-(diethylamino)ethanol hydrochloride is as a pharmacological agent. It has been studied for its potential as a nootropic, a class of substances known to enhance cognitive function. Research has suggested that diethylaminoethanol may promote the synthesis of phosphatidylcholine, a vital component of cell membranes, and contribute to improved memory and learning capabilities. This has led to its use in various dietary supplements aimed at enhancing mental performance. Furthermore, 2-(diethylamino)ethanol hydrochloride is utilized in cosmetic formulations due to its skin-conditioning properties. It is often included in products designed to improve skin hydration and texture. The compound's ability to enhance the permeability of other ingredients also makes it valuable in topical formulations, ensuring effective delivery of active components into the skin. In addition to its applications in pharmacology and cosmetics, diethylaminoethanol hydrochloride serves as an important intermediate in organic synthesis. Its functional groups allow it to participate in various chemical reactions, making it a useful building block in the production of more complex molecules, including pharmaceuticals, agrochemicals, and fine chemicals. Safety considerations for handling 2-(diethylamino)ethanol hydrochloride include standard laboratory practices. While it is generally regarded as safe when used as directed, appropriate precautions should be taken to avoid skin and eye contact, as the compound may cause irritation. In summary, 2-(diethylamino)ethanol hydrochloride is a significant compound in the fields of pharmacology, cosmetics, and organic synthesis. Its unique properties and versatility continue to drive research and development, particularly in the pursuit of cognitive enhancers and effective topical formulations. References 2023. DEAE/Catechol-Chitosan conjugates as bioactive polymers: Synthesis, characterization, and potential applications. Biomacromolecules, 24(1). DOI: 10.1021/acs.biomac.2c01012 2023. Amine-assisted simultaneous CO2 absorption and mineral carbonation: Effect of different categories of amines. Environmental Science & Technology, 57(27). DOI: 10.1021/acs.est.3c01352 2019. Regulating phase separation behavior of a DEEA-TETA biphasic solvent using sulfolane for energy-saving CO2 capture. Environmental Science & Technology, 53(16). DOI: 10.1021/acs.est.9b02787 |
| Market Analysis Reports |