Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Chengdu Hongsheng Sci-Tech Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cdchemical.com | |||
 | +86 (28) 3769-0186 +86 13568974161 | |||
 | +86 (28) 8587-6205 | |||
 | cdchemical@163.com cdchemi@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Sichuan Weibo Science and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.scweibo.com | |||
 | +86 (28) 8757-8473 +86 13880582998 | |||
 | +86 (28) 8757-8473 | |||
 | dudes@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Palchem | France | |||
|---|---|---|---|---|
 | www.palchem.com | |||
 | +33 (3) 2145-2678 | |||
 | +33 (3) 2145-1125 | |||
 | palchem@wanadoo.fr | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| FAR Chemical, Inc. | USA | |||
|---|---|---|---|---|
 | www.far-chemical.com | |||
 | +1 (321) 723-6160 | |||
 | +1 (321) 723-8753 | |||
 | info@far-chemical.com | |||
| Chemical manufacturer since 1983 | ||||
| Boulder Scientific Company | USA | |||
|---|---|---|---|---|
 | www.bouldersci.com | |||
 | +1 (970) 535-4494 | |||
 | +1 (970) 535-4584 | |||
 | sales@bouldersci.com | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organomagnesium |
|---|---|
| Name | Phenylmagnesium bromide |
| Molecular Structure | 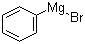 |
| Molecular Formula | C6H5BrMg |
| Molecular Weight | 181.31 |
| CAS Registry Number | 100-58-3 |
| EC Number | 202-867-2 |
| SMILES | C1=CC=[C-]C=C1.[Mg+2].[Br-] |
| Flash point | -40 °C (Expl.) |
|---|---|
| Water solubility | reacts (Expl.) |
| Hazard Symbols |   GHS02;GHS05 Danger Details GHS02;GHS05 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H314 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P264-P280-P301+P330+P331-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P321-P363-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3399 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Phenylmagnesium bromide, a Grignard reagent, is an organomagnesium compound widely used in organic synthesis. It has the chemical formula C6H5MgBr and is characterized by a phenyl group (C6H5) bonded to a magnesium atom, which is further connected to a bromine atom. This reagent, typically found as a solution in ether or tetrahydrofuran (THF), is a cornerstone of organometallic chemistry due to its versatility and reactivity. Phenylmagnesium bromide was discovered in the late 19th century by Victor Grignard during his groundbreaking work on organomagnesium halides. His discoveries earned him the 1912 Nobel Prize in Chemistry and laid the foundation for the extensive use of Grignard reagents in organic synthesis. Grignard's work demonstrated that these compounds could react with a wide variety of electrophiles, enabling the construction of complex molecules from simpler precursors. Phenylmagnesium bromide quickly became one of the most studied and utilized Grignard reagents because of the ubiquity of the phenyl group in chemical and pharmaceutical compounds. The primary application of phenylmagnesium bromide lies in its ability to act as a nucleophile in organic reactions. It is commonly employed in the formation of carbon-carbon bonds, particularly in the addition to carbonyl compounds such as aldehydes, ketones, and esters. These reactions produce secondary or tertiary alcohols, depending on the substrate. For example, the reaction of phenylmagnesium bromide with benzaldehyde yields diphenylmethanol, a secondary alcohol. In addition to alcohol synthesis, phenylmagnesium bromide is utilized in other important transformations. It is an essential reagent in the formation of aromatic ketones via reactions with acyl chlorides or esters. For instance, the reaction of phenylmagnesium bromide with methyl benzoate produces benzophenone, an aromatic ketone used in perfumery and as a UV blocker. Furthermore, it serves in the synthesis of phenyl-substituted compounds, such as biphenyls, which are valuable intermediates in pharmaceutical and material sciences. The versatility of phenylmagnesium bromide also extends to its role in the synthesis of heterocycles and functionalized aromatic compounds. It has been employed in the preparation of various natural products and bioactive molecules. For example, the reagent has been used to introduce phenyl groups into complex frameworks, aiding in the design of pharmaceuticals and agrochemicals. Its ability to form robust bonds with a variety of electrophiles makes it indispensable in medicinal chemistry, where phenylated derivatives are often crucial for drug efficacy. While phenylmagnesium bromide is a highly reactive and valuable reagent, handling it requires precautions. It is sensitive to moisture and air, as the magnesium-halogen bond is highly reactive. Contact with water or atmospheric moisture results in rapid decomposition, forming benzene and magnesium hydroxide. Consequently, the reagent is typically stored and used under inert atmospheres, such as nitrogen or argon, to maintain its stability. In conclusion, phenylmagnesium bromide represents a cornerstone reagent in synthetic organic chemistry, enabling the formation of complex molecular architectures. Its discovery by Victor Grignard marked a significant milestone in chemistry, and its applications continue to evolve in modern research and industry. From pharmaceuticals to materials science, phenylmagnesium bromide remains a versatile and indispensable tool for chemists worldwide. References 1. Synthesis: Grignard, V. (1900). "Sur quelques nouvelles combinaisons organométalliques: Phenylmagnesium bromide." Comptes Rendus de l'Académie des Sciences, 130, 1322-1324. 2. Applications: Kharasch, M. S., et al. (1941). "Grignard reagents in organic synthesis: Phenylmagnesium bromide." Chemical Reviews, 29(3), 463-478. DOI: 10.1021/cr60094a002 3. Review: Silverman, G. S. (2002). "Organomagnesium compounds: Phenylmagnesium bromide." Chemical Society Reviews, 31(5), 261-269. DOI: 10.1039/B104338M |
| Market Analysis Reports |