Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Tyger Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.tygersci.com | |||
 | +1 (609) 434-0144 | |||
 | +1 (609) 434-0143 | |||
 | sales@tygersci.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Alichem Inc. | USA | |||
|---|---|---|---|---|
 | www.alichem.com | |||
 | +1 (858) 987-4788 | |||
 | +1 (858) 987-4799 | |||
 | bella@alichem.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Nanjing Fred Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fredbio.com | |||
 | +86 (25) 8469-6168 | |||
 | Austin@fredchem.cn | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: NJFred01 | |||
 | WhatsApp:+86 17302533743 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| HetCat - Heterocycles & Catalysts | Switzerland | |||
|---|---|---|---|---|
 | www.hetcat.com | |||
 | +41 (61) 363-9868 | |||
 | +41 (61) 363-9869 | |||
 | fallahpour@hetcat.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Pyridine derivative |
|---|---|
| Name | 2-Amino-5-methoxypyridine |
| Molecular Structure | 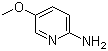 |
| Molecular Formula | C6H8N2O |
| Molecular Weight | 124.14 |
| CAS Registry Number | 10167-97-2 |
| EC Number | 674-589-3 |
| SMILES | COC1=CN=C(C=C1)N |
| Melting point | 36 - 38 °C (Expl.) |
|---|---|
| Boiling point | 251.6±20.0 °C 760 mmHg (Calc.)*, 289.8 - 292.6 °C (Expl.) |
| Flash point | 106.0±21.8 °C (Calc.)* |
| Index of refraction | 1.561 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-Amino-5-methoxypyridine is a nitrogen-containing heterocyclic compound that belongs to the class of substituted pyridines. It has the molecular formula C6H8N2O and consists of a pyridine ring bearing an amino group at the 2-position and a methoxy group at the 5-position. The presence of both electron-donating groups influences the reactivity and physical properties of the molecule, making it particularly useful in synthetic organic chemistry. This compound is frequently employed as an intermediate in the synthesis of pharmaceutical and agrochemical agents. Its structural features allow for multiple derivatization pathways, especially those involving the amino group, which can undergo acylation, sulfonation, or condensation with carbonyl compounds. The methoxy group further enhances the nucleophilic character of the adjacent positions on the aromatic ring, making the molecule suitable for electrophilic substitution reactions and facilitating the construction of more complex heterocyclic systems. 2-Amino-5-methoxypyridine is also used in the development of biologically active molecules. It serves as a key building block for the synthesis of fused heterocycles, such as pyridopyrimidines and pyrazolopyridines, which are often explored for their potential pharmacological properties. The compound has been investigated in the design of enzyme inhibitors, receptor ligands, and antimicrobial agents due to its ability to be incorporated into structures with favorable binding affinities and bioavailability profiles. Synthetically, 2-amino-5-methoxypyridine can be obtained through selective substitution reactions starting from halogenated pyridines or via directed lithiation strategies. Its stability under a range of reaction conditions and its compatibility with various functional group transformations make it a reliable reagent in multistep syntheses. Due to its versatility and well-characterized reactivity, 2-amino-5-methoxypyridine continues to be an important compound in both industrial and academic research focused on the discovery and optimization of new chemical entities. References 2014. 2,2,3-Tribromopropanal as a Versatile Reagent in the Skraup-Type Synthesis of 3-Bromoquinolin-6-ols. Synlett, 25(7). DOI: 10.1055/s-0033-1340670 |
| Market Analysis Reports |