Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| SynQuest Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.synquestlabs.com | |||
 | +1 (386) 462-0788 | |||
 | +1 (386) 462-7097 | |||
 | Sales@synquestlabs.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Ich Biofarm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ichemie.com | |||
 | +86 (571) 2818-6870 | |||
 | +86 (571) 8993-9479 | |||
 | specialchem1@ichemie.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| SobhaBio | India | |||
|---|---|---|---|---|
 | www.sobhabio.com | |||
 | +91 (99) 8988-2177 | |||
 | sobhabio@gmail.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Zhuhai Hairuide Bioscience and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhhairuide.com | |||
 | +86 (756) 778-9199 +86 13326687259 +86 13326687289 +86 15913232160 | |||
 | +86 (756) 778-9099 | |||
 | sales@zhhairuide.com zhengyanhong@zhhairuide.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridazine |
|---|---|
| Name | 3-Amino-6-methoxypyridazine |
| Synonyms | NSC 73703; (6-Methoxypyridazin-3-yl)amine; 6-Methoxypyridazin-3-amine; 3-Methoxy-6-aminopyridazine; 6-Amino-3-methoxypyridazine |
| Molecular Structure | 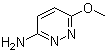 |
| Molecular Formula | C5H7N3O |
| Molecular Weight | 125.13 |
| CAS Registry Number | 7252-84-8 |
| EC Number | 230-670-1 |
| SMILES | COC1=NN=C(C=C1)N |
| Density | 1.2±0.1 g/cm3 Calc.*, 1.224 g/mL (Expl.) |
|---|---|
| Melting point | 108-109 °C (Expl.) |
| Boiling point | 353.5±22.0 °C 760 mmHg (Calc.)* |
| Flash point | 167.6±22.3 °C (Calc.)* |
| Index of refraction | 1.567 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
3-Amino-6-methoxypyridazine is a heterocyclic organic compound featuring a pyridazine ring substituted with an amino group at the 3-position and a methoxy group at the 6-position. The molecular structure consists of a six-membered aromatic ring containing two adjacent nitrogen atoms (pyridazine), which imparts distinct electronic and chemical properties to the compound. The presence of the amino (-NH2) and methoxy (-OCH3) substituents further modifies the reactivity and potential applications of the molecule. The discovery of 3-amino-6-methoxypyridazine can be traced back to investigations of substituted pyridazines, which have attracted significant attention due to their roles as key intermediates in the synthesis of pharmaceuticals, agrochemicals, and functional materials. Pyridazine derivatives have been extensively studied for their bioactive properties, including antibacterial, antifungal, and anti-inflammatory activities. The introduction of electron-donating groups such as amino and methoxy groups into the pyridazine ring framework often enhances these biological effects and improves physicochemical properties like solubility and stability. The synthesis of 3-amino-6-methoxypyridazine typically involves multistep organic reactions starting from substituted pyridazine precursors or through ring-closure methods using appropriate hydrazines and dicarbonyl compounds. One common synthetic route involves the selective substitution of the pyridazine ring at specified positions using nitration, reduction, and methylation reactions to install the amino and methoxy groups. The synthetic accessibility of this compound has facilitated its exploration as a building block in medicinal chemistry and organic synthesis. Applications of 3-amino-6-methoxypyridazine are primarily found in the development of bioactive molecules and functional materials. Its structural features make it a valuable intermediate in the preparation of kinase inhibitors, antimicrobial agents, and other pharmacologically relevant compounds. The amino group allows for further functionalization through acylation, sulfonation, or coupling reactions, enabling the design of molecules with targeted biological activities. In agrochemical research, pyridazine derivatives bearing amino and methoxy substituents have been investigated as potential herbicides and fungicides. The ability of such compounds to interfere with enzymatic pathways in pests and pathogens underlines their importance in crop protection strategies. Moreover, the methoxy group can enhance membrane permeability and metabolic stability, improving the efficacy of active compounds derived from this scaffold. In addition to pharmaceutical and agrochemical uses, 3-amino-6-methoxypyridazine is also employed in materials science, particularly in the design of organic semiconductors and fluorescent probes. The heterocyclic ring and electron-rich substituents contribute to the compound's photophysical properties, making it suitable for incorporation into dyes and sensors. Physicochemically, 3-amino-6-methoxypyridazine exhibits moderate solubility in polar organic solvents such as methanol, ethanol, and dimethyl sulfoxide, while its aqueous solubility is limited due to the aromatic structure. Its melting point and spectral properties have been characterized using standard analytical techniques including nuclear magnetic resonance spectroscopy, infrared spectroscopy, and mass spectrometry, confirming the positions of substitution and purity. The safety profile and toxicity of 3-amino-6-methoxypyridazine are dependent on its applications and formulation. As a chemical intermediate, it is handled with appropriate precautions to minimize exposure. Its derivatives and analogs are subject to evaluation for environmental and biological effects when developed into active pharmaceutical ingredients or agrochemicals. In summary, 3-amino-6-methoxypyridazine is a heterocyclic compound featuring amino and methoxy substituents on a pyridazine ring. Its discovery and synthetic accessibility have made it an important intermediate in the synthesis of pharmaceuticals, agrochemicals, and functional materials. The compound’s chemical properties enable diverse applications ranging from drug development to materials science. References 2002. Reaction of α-Aminoazines with α-Halo Oxo Reagents. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-012-00806 1969. Investigations in the pyridazine series for compounds possessing tuberculostatic activity. Pharmaceutical Chemistry Journal. DOI: 10.1007/bf01151460 1974. Synthesis of thiourea derivatives. Pharmaceutical Chemistry Journal. DOI: 10.1007/bf00771335 |
| Market Analysis Reports |