Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Eastar Chemical Corporation | USA | |||
|---|---|---|---|---|
 | www.eastarchem.com | |||
 | +1 800-898-2436 | |||
 | +1 (877) 898-2436 | |||
 | info@eastarchem.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shandong Dinghao Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sddhpharm.com | |||
 | +86 (0531) 5856-5868 | |||
 | lily.zhou@sddhpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic acid halide |
|---|---|
| Name | Cyclopentylpropionyl chloride |
| Synonyms | 3-Cyclopentylpropionyl chloride |
| Molecular Structure | 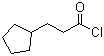 |
| Molecular Formula | C8H13ClO |
| Molecular Weight | 160.64 |
| CAS Registry Number | 104-97-2 |
| EC Number | 203-257-9 |
| SMILES | C1CCC(C1)CCC(=O)Cl |
| Density | 1.049 |
|---|---|
| Boiling point | 199-200 °C |
| Refractive index | 1.464 |
| Flash point | 194 °F |
| Hazard Symbols |  GHS05 Danger Details GHS05 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H318 Details | ||||||||||||||||||||
| Safety Statements | P260-P264-P264+P265-P280-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P363-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 3265 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Cyclopentylpropionyl chloride, a chlorinated organic compound with the molecular formula C8H13ClO, is a derivative of propionic acid where the propionyl group is bonded to a cyclopentane ring. This compound likely emerged during the mid-20th century as part of ongoing research into acyl chlorides and their applications in organic synthesis. The synthesis of cyclopentylpropionyl chloride involves the chlorination of cyclopentylpropionic acid using reagents such as thionyl chloride (SOCl2) or oxalyl chloride ((COCl)2). Its discovery provided chemists with a new reagent for introducing the cyclopentylpropionyl group into various molecular frameworks. Cyclopentylpropionyl chloride is a valuable intermediate in the pharmaceutical industry. Its ability to introduce the cyclopentylpropionyl group into target molecules makes it useful in the synthesis of active pharmaceutical ingredients (APIs). This reactivity is particularly important in the development of drugs where specific acyl groups can significantly impact biological activity and pharmacokinetic properties. For instance, it is used in creating compounds with enhanced lipophilicity, improving their ability to cross cell membranes and increasing their efficacy. In the agrochemical sector, cyclopentylpropionyl chloride is employed to synthesize herbicides, insecticides, and fungicides. The introduction of the cyclopentylpropionyl group can enhance the stability and effectiveness of these compounds. This modification helps in developing agrochemicals that are more potent against pests and diseases, thereby supporting higher agricultural yields and better crop protection. Cyclopentylpropionyl chloride is used in the development of specialized polymers and advanced materials. By acylating various monomers, it helps create polymers with unique properties such as improved thermal stability, resistance to chemicals, and enhanced mechanical strength. These polymers are used in high-performance materials for coatings, adhesives, and composite materials. In organic synthesis, cyclopentylpropionyl chloride serves as a versatile reagent for introducing acyl groups into molecules. It participates in Friedel-Crafts acylation reactions, forming ketones that are key intermediates in the synthesis of various organic compounds. Cyclopentylpropionyl chloride is used in research and development to study acylation reactions and explore new chemical transformations. Its well-defined reactivity provides a reliable model for investigating reaction mechanisms and optimizing conditions for acylation processes. References 2003. Estradiol cypionate. Pharmaceutical Substances, KD-05-0064. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-05-0064 2003. Testosterone cypionate. Pharmaceutical Substances, KD-20-0043. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-20-0043 2022. Amidation of Polymer-Supported 5-Amino-1-(4-methoxybenzyl)-2-oxo-1,2-dihydroquinoline-3-carboxylic Acid. Science of Synthesis, SD-115-01314. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-115-01314 |
| Market Analysis Reports |