Online Database of Chemicals from Around the World
| Institute of Petrochemistry, Heilongjiang Academy of Science | China | |||
|---|---|---|---|---|
 | www.oledchem.cn | |||
 | +86 (451) 8262-3696 +86 13945093324 | |||
 | +86 (451) 8262-3696 | |||
 | oledchem@gmail.com lkiwi2000@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1962 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Jilin Optical and Electronic Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jl-oled.com | |||
 | +86 (431) 8583-4771 | |||
 | +86 (431) 8583-4772 | |||
 | sales1@jl-oled.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Qingdao Kingway Pharmtech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingwaypharm.com | |||
 | +86 (532) 8711-8899 | |||
 | +86 (532) 8411-7788 | |||
 | sales@kingwaypharm.com kingwaypharm@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Luminescence Technology Corp. | Taiwan | |||
|---|---|---|---|---|
 | www.lumtec.com.tw | |||
 | +886 (2) 2697-5600 | |||
 | +886 (2) 2697-5601 | |||
 | sales@lumtec.com.tw | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Pharmaceutical intermediate >> OLED material intermediate |
|---|---|
| Name | 2,3,6,7,10,11-Hexacyano-1,4,5,8,9,12-hexaazatriphenylene |
| Synonyms | Dipyrazino[2,3-f:2',3'-h]quinoxaline-2,3,6,7,10,11-hexacarbonitrile; HAT-CN |
| Molecular Structure | 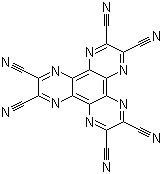 |
| Molecular Formula | C18N12 |
| Molecular Weight | 384.27 |
| CAS Registry Number | 105598-27-4 |
| EC Number | 878-567-6 |
| SMILES | C(#N)C1=C(N=C2C(=N1)C3=NC(=C(N=C3C4=NC(=C(N=C24)C#N)C#N)C#N)C#N)C#N |
| Density | 1.86±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.873, Calc.* |
| Boiling Point | 988.7±65.0 °C (760 mmHg), Calc.* |
| Flash Point | 247.4±19.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P321-P330-P332+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2,3,6,7,10,11-Hexacyano-1,4,5,8,9,12-hexaazatriphenylene, often abbreviated as HAT(CN)6, is a nitrogen-rich aromatic compound known for its exceptional electronic and coordination properties. This molecule was first synthesized in the late 20th century as part of research into polycyano-substituted heterocycles. Its discovery marked a significant advancement in the field of organic materials due to its unique structure, consisting of a hexaazatriphenylene core with six cyano groups attached. This arrangement imparts strong electron-accepting characteristics, making it highly suitable for electronic and optoelectronic applications. The synthesis of 2,3,6,7,10,11-hexacyano-1,4,5,8,9,12-hexaazatriphenylene involves multi-step procedures starting from simple aromatic precursors. One of the most effective methods is the condensation of appropriate diamino and dicyano compounds under controlled conditions, followed by oxidative cyclization to form the hexaazatriphenylene core. The cyano groups are introduced during or after the formation of the aromatic framework. This synthesis method ensures high yield and purity, which is critical for its use in electronic applications. HAT(CN)6 has garnered significant attention in materials science, particularly for its role as an electron acceptor in organic electronics. It has been used in the development of organic field-effect transistors (OFETs), organic photovoltaics (OPVs), and organic light-emitting diodes (OLEDs). Its strong electron-withdrawing properties facilitate efficient charge separation and transport, which are essential for high-performance electronic devices. Additionally, HAT(CN)6 forms stable charge-transfer complexes with various electron donors, making it valuable in the design of conductive and semiconductive materials. Beyond electronics, HAT(CN)6 exhibits potential in coordination chemistry and catalysis. The cyano groups act as coordination sites for metal ions, allowing the formation of metal-organic frameworks (MOFs) with tailored properties. These frameworks are being explored for gas storage, separation, and catalysis. The molecule’s thermal and chemical stability further enhances its suitability for these applications, making it a versatile component in advanced material design. References 2024. Deep-blue organic light-emitting diodes for ultrahigh-definition displays. Nature Photonics, 18(10), 1038-1046. DOI: 10.1038/s41566-024-01508-w 2020. Electrically Conductive 3D Metal-Organic Framework Featuring π-Acidic Hexaazatriphenylene Hexacarbonitrile Ligands with Anion-π Interaction and Efficient Charge-Transport Capabilities. ACS Applied Materials & Interfaces, 12(34), 38416-38425. DOI: 10.1021/acsami.0c12388 2005. Synthesis and mesomorphic behavior of a donor-acceptor-type hexaazatriphenylene. Organic Letters, 7(19), 4261-4264. DOI: 10.1021/ol051231j |
| Market Analysis Reports |