Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhanhua Darong Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.darongchem.com | |||
 | +86 (532) 6777-3200 +86 15689126900 | |||
 | +86 (532) 6777-3200 | |||
 | sales@darongchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou TJM Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjmchem.com | |||
 | +86 13388471767 | |||
 | +86 (571) 8822-3279 | |||
 | sales@tjmchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| IRIS Biotech GmbH | Germany | |||
|---|---|---|---|---|
 | www.iris-biotech.de | |||
 | +49 (9231) 961-973 | |||
 | +49 (9231) 961-999 | |||
 | info@iris-biotech.de | |||
| Chemical manufacturer since 1788 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Thiol/thiophenol |
|---|---|
| Name | p-Toluenethiol |
| Synonyms | Toluene-4-thiol; 4-Methylbenzenethiol; 4-Methylthiophenol; p-Thiocresol |
| Molecular Structure | 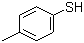 |
| Molecular Formula | C7H8S |
| Molecular Weight | 124.20 |
| CAS Registry Number | 106-45-6 |
| EC Number | 203-399-1 |
| SMILES | CC1=CC=C(C=C1)S |
| Melting point | 40-44 °C |
|---|---|
| Boiling point | 195 °C |
| Flash point | 68 °C |
| Water solubility | slightly soluble |
| Hazard Symbols |    GHS05;GHS06;GHS07 Danger Details GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H302-H311-H312-H315-H318-H319-H330-H331-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P262-P264-P264+P265-P270-P271-P280-P284-P301+P316-P301+P317-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P316-P317-P319-P320-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P403+P233-P405- Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
p-Toluenethiol was first synthesized in the late 19th century as part of early efforts to explore the chemistry of sulfur-containing organic compounds. It was identified and studied for its strong odor and reactive thiol group, becoming a hot topic in understanding thiol chemistry. It is produced by replacing the hydrogen atoms on the methyl group of toluene with thiol groups. p-Toluenethiol is widely used as a reagent in organic synthesis to introduce thiol groups into molecules. Its thiol group is reactive towards various chemical transformations and is valuable for the synthesis of more complex sulfur-containing compounds. p-Toluenethiol is used in the synthesis of thiols, which are important intermediates in the production of pharmaceuticals, agrochemicals, and fragrances. p-Toluenethiol is used to form self-assembled monolayers (SAMs) on metal surfaces, especially gold. The thiol groups in p-toluenethiol form strong bonds with metal atoms to form stable monolayers, thereby changing the surface properties of the substrate. It is also used in surface modification techniques to change the chemical and physical properties of materials for use in sensors, electronics, and catalysis. p-Toluenethiol is used as a heavy metal scavenger in environmental remediation processes. The thiol group has a high affinity for metals, allowing p-toluenethiol to bind and remove heavy metals from contaminated water and soil. p-toluenethiol is used as a metal corrosion inhibitor. Its ability to form a protective film on metal surfaces helps prevent corrosion and degradation in various industrial applications. p-toluenethiol is used for protein and enzyme modification. The thiol group can form disulfide bonds with cysteine residues in proteins, exploring targeted modifications and functions in biochemical studies. It is also being studied for its application as a component of drug delivery systems. The thiol group can form bonds with target molecules or surfaces to enhance the stability and delivery of therapeutic agents. p-toluenethiol is used as a chain transfer agent in the polymerization of vinyl monomers. It helps regulate the molecular weight of the polymer and improve its properties, such as elasticity and thermal stability. p-toluenethiol is used in the formulation of lubricant additives, where the thiol group can react with metal surfaces to provide anti-wear and anti-corrosion properties. p-toluenethiol is used as a lubricant additive, where the thiol group can react with metal surfaces to provide anti-wear and anti-corrosion properties. References 2024. Feedstock chemical dichloromethane as the C1 source for the chemoselective multicomponent synthesis of valuable 1,4,2-dioxazoles. Communications Chemistry, 7(1), 256. DOI: 10.1038/s42004-024-01364-3 2022. Nickel-Catalyzed Regioselective Thiolation of Anilides with Thiols. Synthesis, 54(04), 1081-1088. DOI: 10.1055/s-0041-1737337 2021. Fabrication of La (III) supported on CoFe2O4 MNPs: a novel and efficient heterogeneous catalyst for selective oxidation of sulfides and synthesis of symmetrical disulfides. Research on Chemical Intermediates, 47(12), 5031-5050. DOI: 10.1007/s11164-021-04629-x |
| Market Analysis Reports |