Online Database of Chemicals from Around the World
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Chemceed Corportaion | USA | |||
|---|---|---|---|---|
 | www.chemceed.com | |||
 | +1 (715) 726-2300 | |||
 | +1 (715) 726-2300 | |||
 | inquiry@chemceed.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2009 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Sinyear Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinyear.com | |||
 | +86 (21) 5643-6307 | |||
 | +86 (21) 5168-5846 | |||
 | 42311713@qq.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Chembridge International Corp. Taipei / Dongguan / Nanjing | China | |||
|---|---|---|---|---|
 | www.chembridge.com.tw | |||
 | +886 (2) 2649-6320 +86 (769) 2250-4087 +86 (25) 8471-2192 | |||
 | +886 (2) 2704-3915 / +86 (769) 2246-8225 / +86 (25) 8471-2052 | |||
 | cnservice@chembridge.net | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shanghai Fuqi Industrial & Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fqsh.com | |||
 | +86 (21) 6631-0933 | |||
 | +86 (21) 6631-0933 | |||
 | fqchem@126.com | |||
| Chemical distributor since 2002 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shandong S Sailing Chemical Co., Ltd | China | |||
|---|---|---|---|---|
 | www.ssailing.cn | |||
 | +86 13325053493 | |||
 | qihang@ssailing.cn | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Shandong Near Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nearchemical.net | |||
 | +86 15615461505 | |||
 | belinda@nearchemical.net | |||
 | QQ Chat | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Shandong Aojin Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aojinchem.com | |||
 | +86 18615331147 | |||
 | info@aojinchem.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Nantong Zhonghe Chemical New Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ntzhhg.com | |||
 | +86 13003551299 | |||
 | 2369399482@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Cambridge Isotope Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.isotope.com | |||
 | +1 (978) 749-8000 | |||
 | +1 (978) 749-2768 | |||
 | cilsales@isotope.com | |||
| Chemical manufacturer | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Classification | Organic raw materials >> Alcohols, phenols, phenolic compounds and derivatives >> Acyclic alcohol |
|---|---|
| Name | Ethylene glycol |
| Synonyms | 1,2-Ethanediol; Monoethylene glycol |
| Molecular Structure | 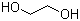 |
| Molecular Formula | C2H6O2 |
| Molecular Weight | 62.07 |
| CAS Registry Number | 107-21-1 |
| EC Number | 203-473-3 |
| SMILES | C(CO)O |
| Density | 1.113 g/mL |
|---|---|
| Melting point | -13 °C |
| Boiling point | 196-198 °C |
| Refractive index | 1.43-1.433 |
| Flash point | 116 °C |
| Water solubility | miscible |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H373 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P264-P270-P301+P317-P319-P330-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Ethylene glycol is an organic compound with the molecular formula C2H6O2, and the structural formula HOCH2CH2OH. It is the simplest member of the glycol family, consisting of two hydroxyl groups attached to a two-carbon alkyl chain. In its pure form, ethylene glycol is a colorless, odorless, sweet-tasting, viscous liquid that is miscible with water and many polar organic solvents. The compound was first prepared in the mid-19th century by the French chemist Charles-Adolphe Wurtz via the hydration of ethylene oxide. Since then, the production process has evolved significantly, and today ethylene glycol is primarily manufactured through the catalyzed hydration of ethylene oxide. This method is widely used in industry due to its efficiency and high product yield. Ethylene glycol is best known for its use as an antifreeze agent in automotive and industrial cooling systems. When mixed with water, it lowers the freezing point and raises the boiling point of the solution, providing protection against extreme temperatures. This property makes it essential for use in internal combustion engine cooling systems in a wide range of climates. Another major application of ethylene glycol is in the production of polyethylene terephthalate (PET), a common plastic used to manufacture beverage bottles, food packaging, and synthetic fibers. In this application, ethylene glycol acts as a monomer in the polymerization process, reacting with terephthalic acid or its dimethyl ester to form PET. The demand for PET has made ethylene glycol a critical feedstock in the global plastics industry. In addition to its roles in antifreeze formulations and PET production, ethylene glycol serves as an intermediate in the manufacture of other chemicals, including unsaturated polyester resins and plasticizers. It is also used as a solvent in various industrial applications and as a dehydrating agent in natural gas processing to remove water vapor. Ethylene glycol is toxic to humans and animals when ingested. Its metabolism in the body leads to the formation of oxalic acid and other harmful compounds that can cause metabolic acidosis, kidney failure, and central nervous system depression. Due to its sweet taste, accidental or intentional ingestion is a known risk, especially for children and pets. As a result, efforts have been made to reduce such risks by adding bittering agents to commercial antifreeze products to deter ingestion. In controlled laboratory settings, ethylene glycol is used in the synthesis of organics, particularly in reactions that require diols or the formation of cyclic acetals and ketals. It is also employed as a cryoprotectant in biological and biochemical research, where it helps protect cells and tissues during freezing. Ethylene glycol has a boiling point of approximately 197 °C and a melting point of about –12.9 °C. It is stable under normal storage conditions but may form explosive peroxides upon prolonged exposure to air and light. Appropriate safety precautions are necessary when handling the substance, especially in high-temperature environments or in confined spaces. The compound remains a vital part of the global chemical industry, with well-established applications in automotive, textile, packaging, and research sectors. Its continued use is supported by extensive literature confirming its chemical behavior, industrial relevance, and health and safety considerations. References 1976. Reaction of hydrogen bromide with diols of long chain α,β-unsaturated acids. Journal of the American Oil Chemists' Society, 53(4). DOI: 10.1007/bf02586346 2021. Methanolic extract of Cucumis melo attenuates ethylene glycol-induced nephrolithiasis in Wistar rats. Urolithiasis, 49(5). DOI: 10.1007/s00240-021-01263-5 2021. Analysis of the polyester clothing value chain to identify key intervention points for sustainability. Environmental Sciences Europe, 33(1). DOI: 10.1186/s12302-020-00447-x |
| Market Analysis Reports |