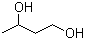
1,3-Butanediol, with the chemical formula C₄H₁₀O₂, is a diol with hydroxyl groups attached to the 1 and 3 positions of a butane chain. This compound is notable for its applications across various industries due to its versatile chemical properties and functional groups.
The discovery of 1,3-butanediol dates back to the early 20th century. It was first synthesized in 1920 by the German chemist Carl Duisberg and his colleagues. The synthesis of 1,3-butanediol involves the hydrogenation of butanedione or the reduction of 1,3-butandione using suitable reducing agents. The compound can be obtained through various methods, including catalytic hydrogenation and the use of specific enzymes.
1,3-Butanediol finds numerous applications in the chemical and pharmaceutical industries. One of its primary uses is as a precursor in the production of polymers. It is a key component in the synthesis of polybutylene terephthalate (PBT), a thermoplastic polymer used in engineering plastics and automotive parts. PBT is valued for its strength, rigidity, and resistance to chemical and thermal degradation, making it suitable for a range of industrial applications.
In addition to its role in polymer production, 1,3-butanediol is used as a solvent and chemical intermediate. It is employed in the manufacture of various chemicals, including plasticizers and surfactants. The compound's solubility and low toxicity make it an ideal candidate for use in a range of formulations, from cleaning agents to personal care products.
The compound is also used in the pharmaceutical industry, where it serves as an intermediate in the synthesis of various drugs and drug delivery systems. Its role in pharmaceutical applications underscores its importance in the development of therapeutic agents and drug formulations.
In the field of biotechnology, 1,3-butanediol is produced through the fermentation of sugars by certain microorganisms. This biotechnological production method offers a sustainable alternative to traditional chemical synthesis and aligns with the growing emphasis on green chemistry and renewable resources.
Overall, 1,3-butanediol's diverse applications and its role as a precursor in polymer production, chemical synthesis, and biotechnology highlight its significance in both industrial and scientific contexts.
References
1994. Assay of the Enantiomers of 1,2-Propanediol, 1,3-Butanediol, 1,3-Pentanediol, and the Corresponding Hydroxyacids by Gas Chromatography-Mass Spectrometry. Analytical Biochemistry.
DOI: 10.1006/abio.1994.1420
1984. In Vitro Hepatic Gluconeogenesis during Experimental Ketosis Produced in Steers by 1,3-Butanediol and Phlorizin. Journal of Dairy Science.
DOI: 10.3168/jds.s0022-0302(84)81574-x
2024. Thermophysical Properties and PC-SAFT Modeling of Binary Mixtures (Glycerol + 1,2-Ethanediol and Glycerol + 1,2-Propanediol) and Ternary Mixtures (Glycerol + Water + 1,2-Ethanediol, Glycerol + Water + 1,2-Propanediol, and Glycerol + Water + 1,3-Butanediol), at Various Temperatures and Atmospheric Pressure. International Journal of Thermophysics.
DOI: 10.1007/s10765-024-03381-9
|














































 GHS02 Warning Details
GHS02 Warning Details