Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Qufu Hongly Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hongly.com | |||
 | +86 (537) 448-1166 449-1986 449-1996 | |||
 | +86 (537) 449-6569 | |||
 | hongly008@gmail.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Yizheng Haifan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yzhaifan.com | |||
 | +86 (514) 8346-8749 | |||
 | +86 (514) 8266-0277 | |||
 | zcw@yzhaifan.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Papchem Lifesciences | India | |||
|---|---|---|---|---|
 | papchemlifesciences.com | |||
 | 9061328401 | |||
 | bd@papchemlifesciences.com | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Organica Feinchemie GmbH | Germany | |||
|---|---|---|---|---|
 | www.organica.de | |||
 | +49 (3494) 63-6215 | |||
 | +49 (3494) 63-6165 | |||
 | dmi@weylchem-organica.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic potassium |
|---|---|
| Name | Potassium phthalimide |
| Synonyms | Phthalimide potassium; Phthalimide potassium salt; N-Potassium phthalimide |
| Molecular Structure | 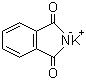 |
| Molecular Formula | C8H4KNO2 |
| Molecular Weight | 185.22 |
| CAS Registry Number | 1074-82-4 |
| EC Number | 214-046-6 |
| SMILES | C1=CC=C2C(=C1)C(=O)[N-]C2=O.[K+] |
| Density | 1.63 g/mL |
|---|---|
| Melting point | 300 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Potassium phthalimide is an important compound with the molecular formula C8H4KNO2. It is the potassium salt of phthalimide, a derivative of phthalic anhydride. Potassium phthalimide is a white crystalline solid that is highly regarded for its reactivity and usefulness in various chemical syntheses. Potassium phthalimide was first synthesized in the late 19th century during the study of phthalic acid derivatives. The compound gained fame in 1888 when Italian chemist Hugo Schiff explored the usefulness of phthalimide in organic synthesis. Schiff's work highlighted the compound's potential for the formation of new amines, paving the way for its widespread use in synthetic organic chemistry. Potassium phthalimide is typically prepared by reacting phthalimide with potassium hydroxide (KOH) in aqueous solution. The product is then isolated by crystallization to yield a pure white crystalline solid. Potassium phthalimide is best known for its role in the Gabriel synthesis, a method for preparing primary amines. In this reaction, potassium phthalimide reacts with an alkyl halide to form an N-alkylphthalimide. The product is then hydrolyzed to produce the primary amine. This method is favored for its specificity and mild reaction conditions, making it ideal for synthesizing amines that are sensitive to harsher conditions. In the pharmaceutical industry, potassium phthalimide is used to synthesize a variety of drug intermediates. Its role in the production of primary amines is particularly important because many drug compounds contain amine groups. The Gabriel synthesis allows for the efficient and selective introduction of these functional groups. Potassium phthalimide is also used in polymer chemistry. It acts as a nucleophile in polymer modification reactions, capable of introducing functional groups into the polymer backbone. This can alter the physical and chemical properties of the polymer, enhancing its performance in a variety of applications such as drug delivery systems, coatings, and adhesives. In addition to the Gabriel synthesis, potassium phthalimide is a versatile organic synthesis reagent. It can participate in a variety of substitution and condensation reactions, facilitating the construction of complex organic molecules. Its reactivity and stability make it a reliable tool for chemists working in the synthesis of fine chemicals and materials. References 2023. Synthesis of New Cyclohexenone Derivatives Using Potassium Phthalimide as a Green Organocatalyst. One-Pot Microwave-Assisted Synthesis and Antimicrobial Evaluation. Russian Journal of Organic Chemistry. DOI: 10.1134/s107042802301013x 2023. N-Phthalimide as a Site-Protecting and Stereodirecting Group in Rhodium-Catalyzed C-H Functionalization with Donor/Acceptor Carbenes. Organic Letters. DOI: 10.1021/acs.orglett.3c00844 2024. Synthesis, carbonic anhydrase inhibition studies and modelling investigations of phthalimide-hydantoin hybrids. Journal of Enzyme Inhibition and Medicinal Chemistry. DOI: 10.1080/14756366.2024.2335927 |
| Market Analysis Reports |