Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Futoh Chemicals Co., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.futoh.co.jp | |||
 | +81 (52) 521-8171 | |||
 | +81 (52) 521-8793 | |||
 | tokoro@futoh.co.jp | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Jubilant Organosys Ltd. | India | |||
|---|---|---|---|---|
 | www.jubl.com | |||
 | +91 (120) 436-1221 | |||
 | puneet_malik@jubl.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tocopharm (Shanghai) Co., Limited | China | |||
|---|---|---|---|---|
 | www.tocopharm.com | |||
 | +86 (21) 3160-8287 +86 13776836200 | |||
 | +86 (21) 6129-2409 | |||
 | info@tocopharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Nanjing Chemical Material Generial Corporation | China | |||
|---|---|---|---|---|
 | www.njchemm.com | |||
 | +86 (25) 5233-7978 8663-0794 8663-8880 | |||
 | +86 (25) 8331-1492 / 8330-4509 | |||
 | 1096481733@qq.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Nanjing Hipower International Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njhipower.com | |||
 | +86 (25) 8473-1076 | |||
 | +86 (25) 8473-1070 | |||
 | fenik828@yahoo.com.cn | |||
| Chemical distributor since 2003 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Anchuan Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.achchem.com | |||
 | +86 (571) 8618-1537 2630-3897 | |||
 | sales@achchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Missyou Chenmical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shmychem.com | |||
 | +86 (21) 5858-3907 | |||
 | +86 (21) 5858-3907 | |||
 | doreen@shmychem.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Lasons India Pvt Ltd | India | |||
|---|---|---|---|---|
 | www.lasons.com | |||
 | +91 9819068483 | |||
 | sale3@lasons.in | |||
| Chemical manufacturer since 1979 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Jiangsu Raien Environmental Protection Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.raien.cn | |||
 | +86 16651336278 | |||
 | 1780189359@qq.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Organica Feinchemie GmbH | Germany | |||
|---|---|---|---|---|
 | www.organica.de | |||
 | +49 (3494) 63-6215 | |||
 | +49 (3494) 63-6165 | |||
 | dmi@weylchem-organica.com | |||
| Chemical manufacturer | ||||
| Shanghai Jinjinle Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jjlchem.com | |||
 | +86 (21) 6610-5180 5291-0829 5291-3935 | |||
 | +86 (21) 3201-0235 | |||
 | jjlchem@jjlchem.com | |||
| Chemical manufacturer | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| True Pharmachem Inc. | USA | |||
|---|---|---|---|---|
 | www.truepharmachem.com | |||
 | +1 (217) 766-6932 | |||
 | +1 (973) 588-3470 | |||
 | sales@truepharmachem.com | |||
| Chemical manufacturer | ||||
| Tengzhou Zhongtai Fine Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhongtaifc.com | |||
 | +86 (632) 265-9369 +86 13906325632 | |||
 | +86 (632) 557-7378 | |||
 | zhongtaifc@163.net | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Classification | Organic raw materials >> Heterocyclic compound |
|---|---|
| Name | 2,6-Lutidine |
| Synonyms | 2,6-Dimethylpyridine |
| Molecular Structure | 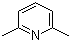 |
| Molecular Formula | C7H9N |
| Molecular Weight | 107.15 |
| CAS Registry Number | 108-48-5 |
| EC Number | 203-587-3 |
| FEMA | 3540 |
| SMILES | CC1=NC(=CC=C1)C |
| Density | 0.9252 |
|---|---|
| Melting point | -6 °C |
| Boiling point | 143-145 °C |
| Refractive index | 1.4966-1.4986 |
| Flash point | 33 °C |
| Water solubility | 40 g/100 mL (20 °C) |
| Hazard Symbols |   GHS02;GHS07 Warning Details GHS02;GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H302-H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1993 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
2,6-Lutidine (C7H9N), also known as 2,6-lutidine, is a heterocyclic organic compound first discovered in coal tar fractions. It belongs to the family of pyridine isomers, characterized by two methyl groups attached to the pyridine ring at the 2 and 6 positions. The discovery of 2,6-lutidine dates back to the late 19th century, when chemists were extensively studying coal tar components to separate and understand various aromatic compounds. 2,6-Lutidine is a colorless liquid with a characteristic pungent odor. It has a boiling point of about 144°C and is slightly soluble in water, but is readily soluble in organic solvents such as ethanol, ether, and chloroform. The presence of methyl groups at the 2 and 6 positions of the pyridine ring makes it less reactive than pyridine itself, but it has unique stereo and electronic properties that are valuable in various chemical reactions. 2,6-Lutidine is widely used as a base and catalyst in organic synthesis. Its relatively low nucleophilicity compared to pyridine makes it an ideal base for reactions that require deprotonation but not nucleophilic attack. It is used as a catalyst in Friedel-Crafts acylation and other reactions that require mild basic conditions to promote carbon-carbon or carbon-heteroatom bond formation. In the pharmaceutical industry, 2,6-lutidine is a valuable reagent and intermediate for the synthesis of active pharmaceutical ingredients (APIs) and intermediates. Its role as a base aids in the formation of a variety of pharmacologically active compounds. It is also used for the protection and deprotection of functional groups during multistep organic synthesis, ensuring that specific reactions occur without interference from other reactive sites in the molecule. 2,6-Lutidine plays a role in polymer chemistry as a catalyst for the polymerization of certain monomers, helping to produce specialty polymers with specific properties. In coordination chemistry, 2,6-lutidine is used as a ligand that is able to donate electron density through the nitrogen atom, making it a useful ligand for the formation of metal complexes. These complexes are studied for their catalytic, magnetic, and electronic properties. 2,6-Lutidine is a staple in chemical research laboratories, and due to its alkalinity and solubility, it is used as an analytical reagent in a variety of chemical assays and titrations. Its unique properties make it a subject of study for the development of new chemical reactions and materials. 2,6-Lutidine is a flammable liquid that can be a health hazard if not handled properly. It can cause skin and eye irritation, and inhalation of its vapors can lead to respiratory irritation. When handling this chemical, appropriate personal protective equipment (PPE) such as gloves, goggles, and adequate ventilation must be worn to minimize the risk of exposure. Although 2,6-Lutidine is not considered to be very toxic to the environment, it should still be properly managed to prevent contamination. Waste disposal and discharge should be controlled according to regulatory guidelines to minimize environmental impact. References 2015. Two-step one-pot synthesis of dihydropyrazinones as Xaa-Ser dipeptide isosteres through morpholine acetal rearrangement. Organic & Biomolecular Chemistry. DOI: 10.1039/c5ob00783f 2013. Synthesis, characterisation and biological activities of [(p-cym)RuX(pz4lut)]n+ and [{(p-cym)RuX}2(μ-pz4lut)]n+ (X = Cl, H2O and pz4lut = α,α,α′,α′-tetra(pyrazol-1-yl)-2,6-lutidine). Dalton Transactions. DOI: 10.1039/c3dt51275d 1953. [The bromination of 2-picoline, 4-chloro-2-picoline and 4-chloro-2,6-lutidine by means of N-bromosuccinimide]. Pharmaceutical Bulletin. DOI: 10.1248/cpb1953.1.293 |
| Market Analysis Reports |