Online Database of Chemicals from Around the World
| Suzhou Eagro Limited | China | |||
|---|---|---|---|---|
 | www.eagro.net | |||
 | +86 (512) 6818-8055 | |||
 | +86 (512) 6818-8066 | |||
 | don@eagro.net | |||
| Chemical distributor since 2004 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Tianjin Kaiyuan Chemical Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kaiyuanchem.com | |||
 | +86 (22) 5981-2778 | |||
 | +86 (22) 5981-2779 | |||
 | sales@kaiyuanchem.com junjinan118@msn.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| KKyemistry (India) Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.kyemistryindia.com | |||
 | +91 (40) 2351-0950 | |||
 | +91 (40) 2351-0667 | |||
 | info@kyemistryindia.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Vesta Intracon Bv | Netherlands | |||
|---|---|---|---|---|
 | www.vestachem.com | |||
 | +31 (38) 422-2577 | |||
 | +31 (38) 422-2566 | |||
 | sales@vestachem.com | |||
| Chemical distributor since 1990 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Ningbo Sinotop Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinotopchem.com | |||
 | +86 (574) 8823-7971 | |||
 | +86 (574) 8823-7323 | |||
 | mia@sinotopchem.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Trigona oHG | Germany | |||
|---|---|---|---|---|
 | www.trigona.de | |||
 | +49 (611) 962-5283 | |||
 | +49 (611) 962-9032 | |||
 | info@trigona.de | |||
| Chemical manufacturer | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Liyang Yutian Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yutianchem.com | |||
 | +86 (519) 8719-0569 | |||
 | +86 (519) 8719-0111 | |||
 | yhh@yutianchem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical reagent >> Organic reagent >> Alkane |
|---|---|
| Name | 4-Methylmorpholine |
| Synonyms | 4-Methyl-1-oxa-4-azacyclohexane; N-Methylmorpholine |
| Molecular Structure | 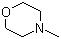 |
| Molecular Formula | C5H11NO |
| Molecular Weight | 101.15 |
| CAS Registry Number | 109-02-4 |
| EC Number | 203-640-0 |
| SMILES | CN1CCOCC1 |
| Density | 0.92 |
|---|---|
| Melting point | -66.1 °C |
| Boiling point | 115-116 °C (750 mmHg) |
| Refractive index | 1.4339-1.4359 |
| Flash point | 13 °C |
| Water solubility | >500 g/L (20 °C) |
| Hazard Symbols |     GHS02;GHS05;GHS07;GHS08 Danger Details GHS02;GHS05;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H302-H312-H314-H318-H332-H373 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P270-P271-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P319-P321-P330-P362+P364-P363-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2535 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
4-Methylmorpholine, with the chemical formula C₅H₁₁NO, is a nitrogen-containing heterocyclic compound featuring a morpholine ring with a methyl group attached at the fourth position. Its discovery and subsequent applications have made it an important compound in various fields, particularly in organic synthesis and chemical processing. The synthesis of 4-methylmorpholine dates back to the early 20th century, as chemists explored modifications of the morpholine ring to enhance its chemical properties and utility. Morpholine itself was first synthesized in 1851 by the German chemist Richard Friedrich August Romain. Over time, researchers sought to modify this structure to improve its reactivity and performance in chemical reactions. The introduction of a methyl group at the fourth position of the morpholine ring led to the development of 4-methylmorpholine, a compound with distinct chemical characteristics compared to its parent molecule. One of the primary applications of 4-methylmorpholine is as a catalyst in organic synthesis. Its basic nature allows it to act as a nucleophilic catalyst in various reactions, such as the formation of imines and the synthesis of heterocyclic compounds. 4-Methylmorpholine is particularly useful in processes involving the activation of electrophiles or the facilitation of nucleophilic substitutions. Its ability to stabilize transition states and intermediate species makes it a valuable tool in the development of new chemical transformations. In addition to its role as a catalyst, 4-methylmorpholine is used as a solvent in certain chemical processes. Its relatively high polarity and ability to dissolve a wide range of organic compounds make it suitable for use in extraction and purification procedures. The solvent properties of 4-methylmorpholine are exploited in the synthesis of various fine chemicals and pharmaceuticals, where it helps to facilitate reaction conditions and enhance product yields. Another significant application of 4-methylmorpholine is in the production of polymeric materials. It is used as a chain transfer agent in the polymerization of certain monomers, where it helps to control molecular weight and improve the properties of the resulting polymers. This application is important in the manufacture of polymers used in coatings, adhesives, and other materials where precise control over polymer structure is required. 4-Methylmorpholine is also utilized in the manufacture of agrochemicals and pharmaceuticals. Its role in chemical synthesis extends to the production of various active pharmaceutical ingredients (APIs) and intermediates, where it serves as a reagent or catalyst in critical reaction steps. In agrochemical formulations, it contributes to the development of compounds with specific biological activities. Handling 4-methylmorpholine requires attention to safety, as it is known to be irritating to the skin, eyes, and respiratory tract. Proper protective measures and safety protocols should be observed to minimize exposure and potential health risks. Overall, the discovery and application of 4-methylmorpholine have significantly contributed to advancements in organic synthesis, polymer chemistry, and industrial processes. Its versatility as a catalyst, solvent, and reagent highlights its importance in the development of various chemical products and materials. References 2016. Imidazolium-based ionic liquids for cellulose pretreatment: recent progresses and future perspectives. Applied Microbiology and Biotechnology. DOI: 10.1007/s00253-016-8057-8 2013. Chemical Derivatization of Peptide Carboxyl Groups for Highly Efficient Electron Transfer Dissociation. Journal of The American Society for Mass Spectrometry. DOI: 10.1007/s13361-013-0701-2 2007. Kinetic Evidence for the Formation of Monocationic N,N'-Disubstituted Phthalamide in Tertiary Amine-Catalyzed Hydrolysis of N-Substituted Phthalimides. The Journal of Organic Chemistry. DOI: 10.1021/jo701295n |
| Market Analysis Reports |