Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| VanDeMark Chemical Inc. | USA | |||
|---|---|---|---|---|
 | www.vandemark.com | |||
 | +1 (716) 433-6764 | |||
 | +1 (716) 433-2850 | |||
 | sales@vandemark.com | |||
| Chemical manufacturer since 1951 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hunan Daochen Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.daochenkj.com | |||
 | +86 18173145536 | |||
 | dashingtech@163.com | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Cn Chemunion Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cnchemunion.com | |||
 | +86 17366116869 | |||
 | david@cnchemunion.com | |||
 | QQ Chat | |||
 | WeChat: +86 15250069576 | |||
 | WhatsApp:+86 17366116869 | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Propyl esters |
|---|---|
| Name | Propyl chloroformate |
| Synonyms | n-Propyl Chloroformate; Carbonochloridic acid propyl ester; Chloroformic acid propyl ester |
| Molecular Structure | 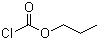 |
| Molecular Formula | C4H7ClO2 |
| Molecular Weight | 122.55 |
| CAS Registry Number | 109-61-5 |
| EC Number | 203-687-7 |
| SMILES | CCCOC(=O)Cl |
| Density | 1.09 |
|---|---|
| Boiling point | 105-106 °C |
| Refractive index | 1.404 |
| Flash point | 84 °F |
| Hazard Symbols |    GHS02;GHS06;GHS05 Danger Details GHS02;GHS06;GHS05 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H331-H314 Details | ||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P270-P271-P280-P284-P301+P317-P301+P330+P331-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P320-P321-P330-P363-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 2740 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Propyl chloroformate is an organic compound with the chemical formula C4H7ClO2. It is a colorless liquid that is primarily used as a reagent in organic synthesis. The chloroformate group, combined with a propyl chain, makes it highly reactive and useful in the formation of esters and carbamates. Propyl chloroformate is a valuable intermediate in producing various chemical compounds, particularly in pharmaceuticals and agrochemicals. The discovery of propyl chloroformate is rooted in the exploration of chloroformates, a class of compounds known for their ability to introduce ester and carbamate functionalities. This reactivity makes them vital in modifying molecules, especially in drug development and peptide synthesis. Propyl chloroformate is specifically known for its use in coupling reactions, where it reacts with alcohols to form esters and with amines to form carbamates. In peptide synthesis, propyl chloroformate is utilized as a coupling reagent to protect or modify amino acids during the formation of peptide bonds. Its ability to react selectively with functional groups in amino acids ensures smooth peptide elongation without unwanted side reactions. The compound's reactivity and versatility in protecting groups during peptide assembly have made it a standard tool in biochemical research. In the pharmaceutical industry, propyl chloroformate is applied in the synthesis of drug intermediates. Its use allows for the efficient formation of key intermediates in active pharmaceutical ingredients, where control over chemical reactivity is crucial. The introduction of ester or carbamate functionalities through propyl chloroformate often improves the stability or bioavailability of drug molecules. Propyl chloroformate is also employed in the agrochemical industry. It participates in the synthesis of carbamate-based pesticides, which are known for their effectiveness in pest control. The ability of propyl chloroformate to form stable carbamates ensures the long-term stability and efficacy of these products in agricultural settings. Overall, propyl chloroformate continues to be a widely used reagent due to its reactivity, especially in esterification and carbamate formation. Its role in the synthesis of complex molecules highlights its importance in both research and industry. References 2019. Amino Acid Analysis in Physiological Samples by GC-MS with Propyl Chloroformate Derivatization and iTRAQ-LC-MS/MS. Methods in Molecular Biology. DOI: 10.1007/978-1-4939-9639-1_14 2019. Novel Molecular Descriptors for the Liquid- and the Gas-Chromatography Analysis of Amino Acids Analogues Derivatized with n-Propyl Chloroformate. Chromatographia. DOI: 10.1007/s10337-019-03767-0 2020. Derivatization. Principles of Forensic Toxicology. DOI: 10.1007/978-3-030-42917-1_12 |
| Market Analysis Reports |