Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Henan Wanxiang Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.58chem.cn | |||
 | +86 (371) 5666-9001 +86 13949088811 | |||
 | +86 (371) 5666-9008 | |||
 | 58chem@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Scientia Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.scientiapharm.net | |||
 | +86 (21) 5430-1573 6172-1573 400-036-1573 | |||
 | +86 (21) 5186-1981 | |||
 | sales@scientiapharm.com scientiapharm@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Anhui Jin'ao Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ahjachem.com | |||
 | +86 (551) 6368-4116 | |||
 | jinao02@ahjachem.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Chemtech Inductry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjchemtech.com | |||
 | +86 (571) 5671-5668 | |||
 | +86 (571) 85423167 | |||
 | info@zjchemtech.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shandong Heyi Gas Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.heyigas.com | |||
 | +86 (546) 236-9006 | |||
 | +86 (546) 236-9003 | |||
 | sales@heyigas.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Beijing Xinaoxunchi Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xinaoxunchi.com | |||
 | +86 (010) 8934-0150 | |||
 | +86 (010) 8934-0150 | |||
 | hdcfssl@sina.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Gelest, Inc. | USA | |||
|---|---|---|---|---|
 | www.gelest.com | |||
 | +1 (215) 547-1015 | |||
 | +1 (215) 547-2484 | |||
 | info@gelest.com | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Ether |
|---|---|
| Name | Boron trifluoride etherate |
| Synonyms | Boron trifluoride ethyl ether |
| Molecular Structure | 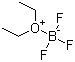 |
| Molecular Formula | C4H10BF3O |
| Molecular Weight | 141.93 |
| CAS Registry Number | 109-63-7 |
| EC Number | 203-689-8 |
| SMILES | B(F)(F)F.CCOCC |
| Density | 1.12 |
|---|---|
| Melting point | -60 °C |
| Boiling point | 126 °C |
| Refractive index | 1.3429-1.3449 |
| Flash point | 48 °C |
| Water solubility | Reacts |
| Hazard Symbols |      GHS02;GHS05;GHS06;GHS07;GHS08 Danger Details GHS02;GHS05;GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H302+H332-H302-H314-H318-H330-H332-H372-H412 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P270-P271-P273-P280-P284-P301+P317-P301+P330+P331-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P319-P320-P321-P330-P363-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2604 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Boron trifluoride etherate, often abbreviated as BF₃·OEt₂, is a coordination complex formed from boron trifluoride (BF₃) and diethyl ether (OEt₂). This compound was developed to stabilize the highly reactive BF₃ gas, making it safer and more practical for use in laboratory and industrial settings. The discovery of boron trifluoride etherate can be traced to early 20th-century research on boron compounds and their reactivity. Boron trifluoride itself was known for its strong Lewis acid properties, but as a gas, it was challenging to handle and transport. Combining BF₃ with diethyl ether created a stable liquid that maintained BF₃’s reactivity while improving its usability, leading to widespread application in organic synthesis and catalysis. The structure of boron trifluoride etherate consists of a central boron atom bonded to three fluorine atoms and coordinated with an ether molecule via the oxygen atom’s lone pair. This coordination is critical, as it stabilizes BF₃ while retaining its potent electron-accepting (Lewis acidic) properties. In this stabilized form, BF₃·OEt₂ is a colorless liquid, soluble in many organic solvents, and easier to store than BF₃ gas. Boron trifluoride etherate is widely used as a catalyst in organic synthesis, particularly in reactions that require strong Lewis acids. It is commonly applied in Friedel-Crafts reactions, where it facilitates the formation of carbon-carbon bonds in the synthesis of aromatic compounds. The compound is also a valuable catalyst in alkylation, acylation, and polymerization reactions, where it activates electrophiles, enhancing the reactivity of organic substrates. In addition, boron trifluoride etherate plays a role in the production of synthetic resins and plastics, where it catalyzes polymerization reactions to form durable materials. The utility of boron trifluoride etherate also extends to analytical chemistry, where it is used in derivatization reactions. In gas chromatography, for instance, it helps convert polar or high-boiling-point substances into volatile derivatives that can be more easily analyzed. This application is critical in the pharmaceutical and environmental sectors, where BF₃·OEt₂ assists in the accurate analysis of complex compounds. The discovery and development of boron trifluoride etherate have provided chemists with a powerful tool for catalysis and synthesis, improving the efficiency and safety of processes involving BF₃. Its applications across organic synthesis, materials science, and analytical chemistry underscore the compound’s importance in both industrial and research environments. References 2024. Fluorination of heterocyclic compounds accompanied by molecular rearrangements. Chemistry of Heterocyclic Compounds. DOI: 10.1007/s10593-024-03340-0 2016. Advances in the synthesis of α-quaternary α-ethynyl α-amino acids. Amino Acids. DOI: 10.1007/s00726-016-2276-2 2009. Stereoselective One-Pot, Three-Component Synthesis of 4-Aryltetrahydropyran via Prins-Friedel-Crafts Reaction. The Journal of Organic Chemistry. DOI: 10.1021/jo802531h |
| Market Analysis Reports |