Online Database of Chemicals from Around the World
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Inhibitor >> Protein tyrosine kinase |
|---|---|
| Name | Tepotinib |
| Synonyms | EMD-1214063; 3-[1,6-Dihydro-1-[[3-[5-[(1-methyl-4-piperidinyl)methoxy]-2-pyrimidinyl]phenyl]methyl]-6-oxo-3-pyridazinyl]benzonitrile |
| Molecular Structure | 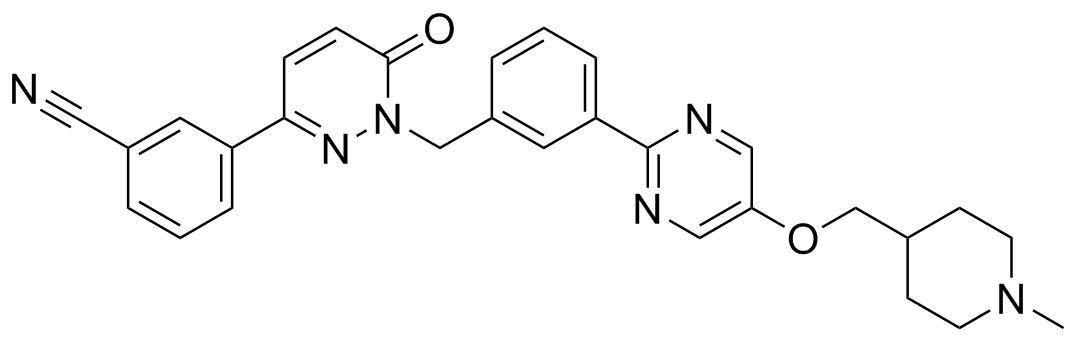 |
| Molecular Formula | C29H28N6O2 |
| Molecular Weight | 492.57 |
| CAS Registry Number | 1100598-32-0 |
| EC Number | 875-266-1 |
| SMILES | CN1CCC(CC1)COC2=CN=C(N=C2)C3=CC=CC(=C3)CN4C(=O)C=CC(=N4)C5=CC=CC(=C5)C#N |
| Density | 1.3$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 626.5$+/-$65.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 332.7$+/-$34.3 $degree$C (Calc.)* |
| Solubility | DMSO 27 mg/mlL (Expl.) |
| Index of refraction | 1.66 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
Tepotinib is a small-molecule targeted anticancer drug developed as a selective inhibitor of the mesenchymal–epithelial transition factor (MET) receptor tyrosine kinase. It is used in oncology for the treatment of non-small cell lung cancer (NSCLC) that harbors MET exon 14 skipping alterations. The compound represents a class of precision medicines designed to inhibit aberrant signaling pathways that drive tumor growth and progression. The development of tepotinib arose from efforts in medicinal chemistry to selectively target MET, a receptor tyrosine kinase involved in cell proliferation, survival, and migration. Under normal physiological conditions, MET is activated by its ligand, hepatocyte growth factor (HGF). In certain cancers, genetic alterations such as exon 14 skipping lead to impaired receptor degradation and prolonged signaling, resulting in oncogenic activation. Tepotinib was designed to inhibit the kinase activity of MET and suppress downstream signaling pathways involved in tumor development. Structurally, tepotinib is a heterocyclic small molecule containing multiple aromatic and heteroaromatic ring systems linked through functionalized side chains. These structural features enable it to bind to the ATP-binding pocket of the MET kinase domain. The binding interaction prevents ATP from accessing the active site, thereby inhibiting phosphorylation events required for receptor activation and signal transduction. The compound exhibits high selectivity for MET over many other kinases, which is important for minimizing off-target effects. The discovery of tepotinib was based on structure-guided drug design and optimization of kinase inhibitor scaffolds. Researchers systematically modified chemical frameworks to improve binding affinity, selectivity, and pharmacokinetic properties. The resulting molecule demonstrated potent inhibition of MET signaling in preclinical models, including suppression of tumor cell proliferation and reduction of metastatic potential. Tepotinib is administered orally and is absorbed into systemic circulation, where it distributes to tumor tissues expressing activated MET. It undergoes hepatic metabolism, primarily involving cytochrome P450 enzymes, and is eliminated through both fecal and renal routes. Its pharmacokinetic profile supports once-daily dosing in clinical use. Clinically, tepotinib is approved for the treatment of metastatic non-small cell lung cancer with MET exon 14 skipping mutations. These genetic alterations lead to abnormal MET signaling that drives tumor growth independent of other oncogenic pathways. By selectively inhibiting MET, tepotinib provides a targeted therapeutic option for patients with this specific molecular subtype of lung cancer. Its use is based on molecular diagnostic testing that identifies eligible patients. The clinical development of tepotinib included studies that evaluated tumor response rates, progression-free survival, and safety profiles in patients with advanced NSCLC. These studies demonstrated meaningful antitumor activity in MET-altered cancers, leading to regulatory approval in multiple regions. The drug is typically used in patients who have limited treatment options due to the presence of specific driver mutations. Adverse effects associated with tepotinib are related to its pharmacological inhibition of MET signaling in normal tissues and include peripheral edema, gastrointestinal disturbances, and liver enzyme elevations. These effects are generally manageable with dose adjustments or supportive care. Overall, tepotinib is a targeted kinase inhibitor designed to selectively block MET receptor signaling in cancers driven by MET exon 14 skipping alterations. Its discovery is rooted in rational drug design focused on kinase inhibition, and its clinical application represents an example of precision oncology, where treatment is tailored to specific genetic alterations in tumors. References 2026. Antisense oligonucleotide targeting TARDBP-EGFR splicing axis inhibits progression of oral squamous cell carcinoma through ABCA1-regulated cholesterol efflux. International Journal of Oral Science. DOI: 10.1038/s41368-025-00402-7 2026. Transcriptomic and multi-layer variant analysis identifies STAT3 and HIF1A as central regulators of regulated cell death pathways in lung squamous cell carcinoma. Naunyn-Schmiedeberg's Archives of Pharmacology. DOI: 10.1007/s00210-026-05103-4 2026. Atrial fibrillation associated with tyrosine kinase inhibitors: A case–control study of real-world pharmacovigilance data. Naunyn-Schmiedeberg's Archives of Pharmacology. DOI: 10.1007/s00210-026-05078-2 |
| Market Analysis Reports |