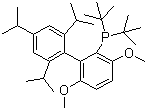
Di-tert-butyl(2',4',6'-triisopropyl-3,6-dimethoxybiphenyl-2-yl)phosphine is a bulky electron-rich phosphine ligand extensively used in modern organic and organometallic chemistry. The compound was discovered as part of efforts to develop highly efficient ligands for catalysis, particularly in the context of palladium-catalyzed cross-coupling reactions. The unique structural features of this phosphine are characterized by the steric hindrance provided by the di-tert-butyl and triisopropyl groups, along with the electron-donating methoxy substituents on the biphenyl backbone. This design allows for stabilization of the metal center while also enabling enhanced reactivity and selectivity in various catalytic processes.
The synthesis of di-tert-butyl(2',4',6'-triisopropyl-3,6-dimethoxybiphenyl-2-yl)phosphine typically involves the introduction of phosphine groups onto pre-functionalized biphenyl precursors. One of the most established methods for synthesizing such ligands begins with the lithiation of a triisopropyl-substituted biphenyl compound followed by the addition of a chlorophosphine reagent such as di-tert-butylchlorophosphine. This reaction proceeds smoothly under anhydrous conditions and leads to the desired phosphine ligand in good yields. Purification can be achieved through standard chromatographic techniques or recrystallization, depending on the specific reaction conditions.
This phosphine ligand has found significant applications in catalysis, particularly in palladium-catalyzed reactions such as Suzuki-Miyaura cross-coupling, Buchwald-Hartwig amination, and other carbon-heteroatom bond-forming processes. Its bulky nature provides excellent stability to the catalytic species while promoting efficient turnover and minimizing unwanted side reactions. The electron-rich environment created by the methoxy substituents enhances the electron density at the metal center, facilitating oxidative addition and reductive elimination steps. These features make it highly suitable for challenging transformations, including coupling reactions involving sterically demanding substrates or electron-poor electrophiles.
In addition to palladium chemistry, this ligand has also shown utility in nickel and copper-catalyzed processes, extending its versatility across a range of synthetic methodologies. Its robustness and high efficiency have contributed to the development of new pharmaceuticals, agrochemicals, and advanced materials. As research into ligand design continues to evolve, di-tert-butyl(2',4',6'-triisopropyl-3,6-dimethoxybiphenyl-2-yl)phosphine remains an important tool for chemists seeking to optimize catalytic reactions for industrial and academic applications.
References
2022. Recent Advances in Metal-Catalyzed Methods for Thioether Synthesis. Synthesis.
DOI: 10.1055/a-1914-1231
2019. Pd-Catalyzed Reactions Promoted by Biaryl Phosphorus Ligands. ACS CatalysisDOI: 10.1021/acsatal.9b03012
2012. By Arylation of aryl ureas. Science of Synthesis
DOI:
|































 GHS07 Warning Details
GHS07 Warning Details