Online Database of Chemicals from Around the World
| Shanghai RC Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rcc.net.cn | |||
 | +86 (21) 5032-2175 5866-1250 ex 603 | |||
 | +86 (21) 5032-2176 / 5866-1251 | |||
 | sales@rcc.net.cn chad@rcc.net.cn | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Jiangsu Ever Galaxy Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.egchemical.com | |||
 | +86 (25) 8459-6982 +86 18602515822 | |||
 | +86 (527) 8445-0055 | |||
 | info@egchemical.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Liaoning Hongtai Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lnhtbio.com | |||
 | +86 (24) 7882-6666 | |||
 | +86 (24) 7882-6999 | |||
 | hongtai.bio@vip.163.com | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Valiant Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.valiant-cn.com | |||
 | +86 (535) 610-1878 | |||
 | +86 (535) 638-6989 | |||
 | jiangyanan@valiant-cn.com chenxiaowei@valiant-cn.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Skyrun Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinaskyrun.com | |||
 | +86 (571) 8672-2205 | |||
 | sales@chinaskyrun.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Yantai Hongbang Pharm Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ythbpharm.com | |||
 | +86 13723949135 | |||
 | yongylmli@126.com | |||
| Chemical distributor since 2018 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Fluorenes |
|---|---|
| Name | Bisphenoxyethanolfluorene |
| Synonyms | 4,4'-(9-Fluorenylidene)bis(2-phenoxyethanol); 9,9-Bis[4-(2-hydroxyethoxy)phenyl]fluorene; BPEF |
| Molecular Structure | 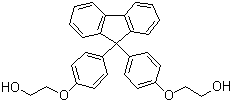 |
| Molecular Formula | C29H26O4 |
| Molecular Weight | 438.51 |
| CAS Registry Number | 117344-32-8 |
| EC Number | 672-704-1 |
| SMILES | C1=CC=C2C(=C1)C3=CC=CC=C3C2(C4=CC=C(C=C4)OCCO)C5=CC=C(C=C5)OCCO |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Bisphenoxyethanolfluorene, also known by its IUPAC name 9,9-bis[4-(2-hydroxyethoxy)phenyl]fluorene, is an organic compound characterized by a fluorene core substituted at the 9-position with two 4-(2-hydroxyethoxy)phenyl groups. This compound belongs to a broader class of materials derived from fluorene structures, which are widely used in polymer chemistry due to their rigidity, thermal stability, and excellent optical properties. The synthesis of bisphenoxyethanolfluorene typically involves the nucleophilic substitution of 9,9-bis(4-hydroxyphenyl)fluorene with ethylene oxide or other ethylene glycol derivatives under basic conditions, resulting in the introduction of hydroxyethoxy side chains. This modification imparts greater flexibility and hydrophilicity to the molecule compared to its parent compound. Bisphenoxyethanolfluorene is primarily used as a monomer or comonomer in the production of high-performance polymers, especially polyesters, polycarbonates, and epoxy resins. Its incorporation into polymeric systems enhances thermal stability and glass transition temperatures due to the rigid and planar fluorene moiety. The hydroxyethoxy groups contribute to improved solubility and processability of the resulting polymers, as well as facilitating cross-linking reactions in thermosetting systems. In the context of electronic materials, bisphenoxyethanolfluorene derivatives have been studied for use in optoelectronic applications such as organic light-emitting diodes (OLEDs), organic field-effect transistors (OFETs), and photovoltaic devices. The fluorene backbone contributes to high charge carrier mobility and efficient light emission, while the hydroxyl functionality allows for further chemical derivatization or network formation in device fabrication. The compound has also been used in the development of flame-retardant polymers. The aromatic and ether linkages, combined with the hydroxyl groups, allow for effective incorporation into phosphorus-containing flame-retardant systems or for reaction with halogenated compounds to enhance thermal and fire resistance of polymeric materials. Bisphenoxyethanolfluorene is generally handled in research and industrial settings under standard safety precautions. It is considered a low-volatility compound but should be handled with care to avoid skin or eye contact due to its phenolic ether structure. It should be stored in a dry, cool environment, away from strong acids or oxidizing agents. Overall, bisphenoxyethanolfluorene represents a valuable building block in the field of polymer science and materials engineering, especially in applications requiring high performance, thermal stability, and tunable solubility characteristics. References |2010. 9,9-Bis[4-(2-chloroethoxy)phenyl]-9H-fluorene. Acta crystallographica. Section E, Structure reports online, 66(9). DOI: 10.1107/s1600536810032046 1998. Bisphenols That Stimulate Cells To Release Alkali Metal Cations: A Structure-Activity Study. Journal of Medicinal Chemistry, 41(21). DOI: 10.1021/jm980315d 2024. Preparation method of diether fluorene based on continuous flow reaction. CN-119019232-A. |
| Market Analysis Reports |