Online Database of Chemicals from Around the World
| Guangzhou Congen Pharmatec Co., Ltd. | China | |||
|---|---|---|---|---|
 | congenpharm.com | |||
 | +86 13380081396 | |||
 | +86 13380081396 | |||
 | marketing@congenpharm.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: Shine_Huang2014 | |||
 | WhatsApp:+8613242332900 | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Pharmaceutical intermediate >> API intermediate |
|---|---|
| Name | tBuO-Ara-Glu(AEEA-AEEA-OH)-OtBu |
| Synonyms | (S)-22-(tert-Butoxycarbonyl)-45,45-dimethyl-10,19,24,43-tetraoxo-3,6,12,15,44-pentaoxa-9,18,23-triazahexatetracontanoic acid |
| Molecular Structure | 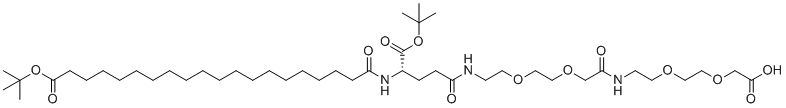 |
| Molecular Formula | C45H83N3O13 |
| Molecular Weight | 874.15 |
| CAS Registry Number | 1188328-37-1 |
| SMILES | CC(C)(C)OC(=O)CCCCCCCCCCCCCCCCCCC(=O)N[C@@H](CCC(=O)NCCOCCOCC(=O)NCCOCCOCC(=O)O)C(=O)OC(C)(C)C |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.483, Calc.* |
| Boiling Point | 953.8±65.0 °C (760 mmHg), Calc.* |
| Flash Point | 530.6±34.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319 Details |
| Safety Statements | P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313 Details |
| SDS | Available |
|
tBuO-Ara-Glu(AEEA-AEEA-OH)-OtBu is a multifunctional compound engineered for advanced applications in drug delivery and biomaterials. The molecule integrates a t-butyl-protected arabinose derivative with a glutamic acid moiety, functionalized by AEEA (aminoethoxyethanol) spacers. This design provides a balance of hydrophobic, hydrophilic, and reactive properties, making it versatile for bioconjugation and material science applications. The development of this compound stems from the need for tailored molecules capable of addressing challenges in targeted drug delivery. The hydrophobic t-butyl-protected arabinose structure ensures stability in complex synthetic pathways, while the glutamic acid core offers multiple functionalization points. The AEEA spacers enhance water solubility and flexibility, which are crucial for applications involving conjugation with large biomolecules like peptides or proteins. Synthesis of tBuO-Ara-Glu(AEEA-AEEA-OH)-OtBu typically involves sequential coupling reactions. Arabinose derivatives are modified with protecting groups to retain stability during subsequent steps. The glutamic acid core is linked through esterification reactions, and AEEA units are incorporated via amide coupling. The t-butyl groups are retained throughout the synthesis to protect the carboxyl functionalities, ensuring controlled reactivity in downstream processes. Applications of this compound are diverse. In drug delivery systems, it serves as a building block for constructing amphiphilic carriers such as micelles and liposomes. These carriers enhance the solubility and bioavailability of hydrophobic drugs. The AEEA spacers also enable functionalization with targeting ligands or therapeutic agents, facilitating precision medicine approaches. In the field of biomaterials, tBuO-Ara-Glu(AEEA-AEEA-OH)-OtBu contributes to the design of biocompatible polymers. Its versatile structure allows for integration into hydrogels, coatings, and scaffold materials used in tissue engineering. The compound’s ability to respond to environmental stimuli, such as pH changes, further expands its utility in controlled drug release systems. This molecule exemplifies the convergence of chemical ingenuity and biomedical innovation, offering a foundation for developing advanced therapeutic and diagnostic technologies. References none |
| Market Analysis Reports |