Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Cangzhou Goldlion Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.goldlionchem.com | |||
 | +86 13315721466 +86 (317) 530-4268 | |||
 | +86 (317) 530-4268 | |||
 | info@goldlionchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Cangzhou Senary Chemical S & T Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.senary.com | |||
 | +86 (317) 548-9300 | |||
 | +86 (317) 548-9300 | |||
 | sale01@senary.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2008 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Henan Ouber Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oubertec.com | |||
 | +86 (371) 6532-2607 +86 18937141980 | |||
 | +86 (371) 6532-2607 | |||
 | anna.zhang@oubertec.com | |||
 | QQ Chat | |||
 | WeChat: 18937141980 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Wudi Reaction Pharma & Chemical Co. Ltd. | China | |||
|---|---|---|---|---|
 | www.ruixinchem.com | |||
 | +86 (0543) 225-7986 +86 15306493269 | |||
 | sales@ruixinchem.com linacaiwu@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Synthetic anti-infective drugs >> Sulfonamides and synergists |
|---|---|
| Name | Glimepiride sulfonamide |
| Synonyms | 4-[2-[(3-Ethyl-4-methyl-2-oxo-3-pyrrolin-1-yl)carboxamido]ethyl]benzenesulfonamide |
| Molecular Structure | 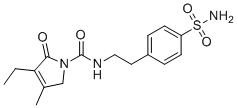 |
| Molecular Formula | C16H21N3O4S |
| Molecular Weight | 351.42 |
| CAS Registry Number | 119018-29-0 |
| EC Number | 601-578-2 |
| SMILES | CCC1=C(CN(C1=O)C(=O)NCCC2=CC=C(C=C2)S(=O)(=O)N)C |
| Solubility | 42.3 g/L (30 °C) (water) (Expl.) |
|---|---|
| Density | 1.3±0.1 g/cm3, Calc.* |
| Melting point | 174.6 °C (Expl.) |
| Index of Refraction | 1.588, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H412 Details | ||||||||
| Safety Statements | P273-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
Glimepiride sulfonamide is a derivative of the widely used antidiabetic drug glimepiride, belonging to the sulfonylurea class of compounds. This chemical entity retains the sulfonamide functionality, which plays a crucial role in its biological activity, and serves as a foundation for potential therapeutic applications and research in medicinal chemistry. The discovery of glimepiride sulfonamide stems from the broader development of sulfonylureas, first synthesized in the 1940s as antibiotics. Their hypoglycemic effects were later recognized, leading to the emergence of sulfonylurea drugs for managing type 2 diabetes. Glimepiride, introduced in the 1990s, represented a third-generation sulfonylurea with improved potency and reduced risk of adverse effects. Sulfonamide derivatives of glimepiride were subsequently explored for their unique properties and potential applications beyond traditional glucose regulation. Glimepiride sulfonamide retains the hypoglycemic action characteristic of its parent compound. It acts by stimulating pancreatic β-cells to secrete insulin, a mechanism mediated by binding to sulfonylurea receptors on the ATP-sensitive potassium channels. This insulinotropic activity positions glimepiride sulfonamide as a candidate for further study in diabetes management, particularly in individuals with partial insulin deficiency. Beyond its established use in glycemic control, glimepiride sulfonamide has drawn attention in the realm of drug design and pharmacology. Researchers have evaluated its structure-activity relationships to develop analogs with enhanced pharmacokinetic profiles or targeted therapeutic properties. The presence of the sulfonamide moiety enables interactions with diverse biological targets, broadening its scope for application in non-diabetic conditions. In oncology, sulfonamide-based compounds have demonstrated anti-proliferative and anti-angiogenic effects. Early-stage research suggests that glimepiride sulfonamide derivatives may inhibit cancer cell growth through pathways unrelated to insulin secretion, such as modulation of cell cycle regulators or apoptotic signaling. These findings provide a promising avenue for further exploration in cancer therapy. Another emerging area of interest is anti-inflammatory activity. The sulfonamide group is a well-documented pharmacophore in anti-inflammatory drugs, and glimepiride sulfonamide has shown potential in reducing inflammation-associated markers in preclinical models. These properties could facilitate its application in conditions characterized by chronic inflammation, such as rheumatoid arthritis or metabolic syndrome. The compound also holds promise in pharmaceutical formulation science. Sulfonamide derivatives often exhibit improved solubility and stability profiles, enhancing bioavailability in drug delivery systems. Glimepiride sulfonamide could thus serve as a model compound for developing advanced formulations to optimize therapeutic efficacy. Despite its potential, challenges remain in the development of glimepiride sulfonamide. Like other sulfonylureas, it carries a risk of hypoglycemia, necessitating careful dose optimization. Additionally, long-term studies are required to assess its safety and efficacy in non-diabetic indications. Glimepiride sulfonamide exemplifies the versatility of sulfonylurea derivatives in both established and experimental therapeutic contexts. Its exploration continues to shed light on innovative applications that extend beyond traditional glucose regulation, paving the way for novel interventions in medicine and pharmacology. References 2017. An Efficient and Practical Process for the Synthesis of Glimepiride. Synlett. DOI: 10.1055/s-0036-1590836 2006. 4-[2-(3-Ethyl-4-methyl-2-oxo-3-pyrrolidine-1-carboxamido)ethyl]benzenesulfonamide. Acta Crystallographica Section E Structure Reports Online. DOI: 10.1107/s1600536806021660 2003. Glimepiride. Pharmaceutical Substances. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-07-0028 |
| Market Analysis Reports |