Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou TJM Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjmchem.com | |||
 | +86 13388471767 | |||
 | +86 (571) 8822-3279 | |||
 | sales@tjmchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Aromsyn Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aromsyn.com | |||
 | +86 (571) 8558-5865 +86 13336024896 | |||
 | +86 (571) 8558-5865 | |||
 | service@aromsyn.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| LOBA Feinchemie AG | Austria | |||
|---|---|---|---|---|
 | www.loba.co.at | |||
 | +43 (223) 277-391 | |||
 | +43 (223) 276-677 | |||
 | sales@loba.co.at | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Bondchemistry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bondchemistry.com | |||
 | +86 15858185835 | |||
 | sales@bondchemistry.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2020 | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Thiol/thiophenol |
|---|---|
| Name | 4-Aminothiophenol |
| Synonyms | 4-Aminobenzenethiol; 4-Mercaptoaniline |
| Molecular Structure | 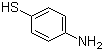 |
| Molecular Formula | C6H7NS |
| Molecular Weight | 125.19 |
| CAS Registry Number | 1193-02-8 |
| EC Number | 214-763-4 |
| SMILES | C1=CC(=CC=C1N)S |
| Melting point | 37-43 °C |
|---|---|
| Boiling point | 140-145 °C (16 mmHg) |
| Flash point | 113 °C |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H314 Details | ||||||||||||||||||||
| Safety Statements | P260-P264-P270-P280-P301+P316-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P321-P330-P363-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 3263 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
4-Aminothiophenol, also known as 4-mercaptoaniline, is a chemical compound with the molecular formula C6H7NS. It is characterized by its aromatic ring structure in which an amino group (-NH2) and a thiol group (-SH) are attached to a thiophene ring. 4-Aminothiophenol is a pale yellow to brown solid at room temperature and is slightly soluble in water, but soluble in organic solvents such as ethanol and acetone. 4-Aminothiophenol was first synthesized and characterized for its chemical properties and structure in organic chemistry research. Its discovery stemmed from the study of thiol-containing aromatic compounds and their reactivity in various chemical reactions. 4-Aminothiophenol is a widely used building block in organic synthesis, especially in the preparation of dyes, pharmaceuticals, and fine chemicals. Its amino and thiol functional groups enable it to participate in various reactions, including nucleophilic substitutions, redox reactions, and metal complex formation. Due to its chemical reactivity and ability to form stable complexes with metals, 4-aminophenol is useful as a precursor in the synthesis of dyes and pigments. It aids in the production of colorants used in textiles, paints, and printing inks. In biological and biomedical research, 4-aminophenol is used as a chemical probe and modifier due to its ability to bind and modify proteins and enzymes through its thiol group. This property makes it valuable in studies involving protein structure-function relationships and enzyme kinetics. The thiol group of 4-aminophenol is used in analytical chemistry to detect and quantify metal ions. It forms stable complexes with metals such as mercury, lead, and copper, enabling sensitive detection methods such as colorimetric assays and electrochemical sensors. 4-Aminothiophenol is toxic when ingested, inhaled, or in contact with the skin and should therefore be handled with care. Appropriate safety precautions must be taken when handling this compound, including the use of gloves, goggles, and adequate ventilation. References 1928. Ü ber Aminothiophenolderivate. Monatshefte für Chemie und verwandte Teile anderer Wissenschaften. DOI: 10.1007/bf01518397 2024. Toward coupling across inorganic/organic hybrid interfaces: polyaniline-coated gold nanoparticles with 4-aminothiophenol as gold-anchoring moieties. Colloid and Polymer Science. DOI: 10.1007/s00396-024-05262-x 2024. Selective deposition of a MOF at the spikes of Au nanostars for SERS detection. Nano Research. DOI: 10.1007/s12274-024-6737-8 |
| Market Analysis Reports |