Online Database of Chemicals from Around the World
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Enki Biopharmaceuticals (Shanghai) Limited | China | |||
|---|---|---|---|---|
 | www.enkibiopharma.com | |||
 | +86 (21) 5768-0965 +86 13916707528 | |||
 | +86 (21) 5768-0922 | |||
 | info@enkibiopharma.com | |||
 | QQ Chat | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Guangdong Raffles Pharma Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rafflespt.com | |||
 | +86 (752) 553-8396 | |||
 | +86 (752) 531-9394 | |||
 | sales4@rafflespt.com | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2017 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Inhibitor >> Mitogen-activated protein kinase (MAPK) >> Raf inhibitor |
|---|---|
| Name | Dabrafenib |
| Synonyms | N-[3-[5-(2-Amino-4-pyrimidinyl)-2-(tert-butyl)-4-thiazolyl]-2-fluorophenyl]-2,6-difluorobenzenesulfonamide |
| Molecular Structure | 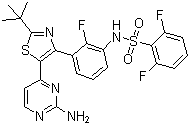 |
| Molecular Formula | C23H20F3N5O2S2 |
| Molecular Weight | 519.56 |
| CAS Registry Number | 1195765-45-7 |
| EC Number | 689-166-9 |
| SMILES | CC(C)(C)C1=NC(=C(S1)C2=NC(=NC=C2)N)C3=C(C(=CC=C3)NS(=O)(=O)C4=C(C=CC=C4F)F)F |
| Solubility | >80 mg/mL (DMSO), <1 mg/mL (water) |
|---|---|
| Density | 1.443 |
| Hazard Symbols |   GHS08;GHS09 Warning Details GHS08;GHS09 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H361f-H373-H400-H401 Details | ||||||||||||||||||||||||
| Safety Statements | P203-P260-P273-P280-P318-P319-P391-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Dabrafenib is a targeted cancer therapy discovered by GlaxoSmithKline in the early 2000s. It emerged from research focused on finding inhibitors of the BRAF kinase, a protein involved in cell growth and division, which is often mutated in various cancers, most notably melanoma. The most common mutation, BRAF V600E, leads to uncontrolled cell proliferation. After extensive preclinical studies and clinical trials demonstrating significant efficacy and manageable safety profiles, dabrafenib received FDA approval in 2013 for the treatment of metastatic melanoma with BRAF V600E mutations. This discovery marked a significant advancement in the development of personalized cancer therapies, offering new hope for patients with specific genetic profiles. Dabrafenib is primarily used in the treatment of melanoma with BRAF V600E mutations. As a BRAF inhibitor, it works by selectively targeting and inhibiting the mutant BRAF kinase, thereby interrupting the aberrant signaling pathways that promote cancer cell growth. This targeted mechanism makes dabrafenib particularly effective in shrinking tumors and controlling disease progression in patients with BRAF-mutant melanoma. In clinical practice, dabrafenib is often used in combination with trametinib, a MEK inhibitor. This combination therapy has been shown to improve survival rates and delay disease progression compared to dabrafenib alone. The synergy between dabrafenib and trametinib helps overcome resistance that often develops with single-agent therapy, providing a more durable response. Beyond melanoma, dabrafenib's applications extend to other cancers harboring BRAF V600E mutations. It has shown efficacy in treating non-small cell lung cancer (NSCLC) and anaplastic thyroid cancer, expanding its therapeutic potential. The approval of dabrafenib for these indications has been a significant step forward in the management of these cancers, offering new options for patients with limited treatment choices. Ongoing research is exploring the use of dabrafenib in combination with other targeted therapies and immunotherapies to further enhance its effectiveness and broaden its applicability. Additionally, studies are investigating biomarkers that can predict response to dabrafenib, aiming to personalize treatment and maximize therapeutic benefits. The development of dabrafenib highlights the importance of precision medicine in oncology. By targeting specific genetic mutations, dabrafenib provides a tailored approach to cancer treatment, improving outcomes and offering new hope to patients with BRAF-mutant cancers. As research continues, dabrafenib's role in cancer therapy is expected to expand, contributing to more effective and personalized treatment strategies. References 2013. Phase II Trial (BREAK-2) of the BRAF Inhibitor Dabrafenib (GSK2118436) in Patients With Metastatic Melanoma. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. DOI: 10.1200/jco.2013.49.8691 2015. Redifferentiation of Iodine-Refractory BRAF V600E-Mutant Metastatic Papillary Thyroid Cancer with Dabrafenib. Clinical cancer research : an official journal of the American Association for Cancer Research. DOI: 10.1158/1078-0432.ccr-14-2915 2018. Cell-free DNA in cancer: current insights. Cellular oncology (Dordrecht, Netherlands). DOI: 10.1007/s13402-018-0413-5 |
| Market Analysis Reports |