Online Database of Chemicals from Around the World
| Shandong Benrite New Chemical Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.benritechem.com | |||
 | +86 (0533) 286-6658 | |||
 | sales@benritechem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: benritechem | |||
 | WhatsApp:+8613021512891 | |||
| Chemical distributor since 2024 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | Phenyl 1H-imidazole-1-sulfonate |
| Synonyms | phenyl imidazole-1-sulfonate |
| Molecular Structure | 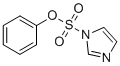 |
| Molecular Formula | C9H8N2O3S |
| Molecular Weight | 224.24 |
| CAS Registry Number | 1198183-95-7 |
| SMILES | C1=CC=C(C=C1)OS(=O)(=O)N2C=CN=C2 |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.619, Calc.* |
| Boiling Point | 400.1±28.0 °C (760 mmHg), Calc.* |
| Flash Point | 195.8±24.0 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
|
Phenyl 1H-imidazole-1-sulfonate is a synthetic compound characterized by a phenyl group attached to an imidazole moiety via a sulfonate linkage. This structure makes it an effective reagent in organic synthesis, particularly in the fields of pharmaceuticals and materials chemistry. The sulfonate functional group provides reactivity that allows the compound to act as a versatile intermediate or coupling agent in the formation of more complex molecules. The discovery of phenyl 1H-imidazole-1-sulfonate can be traced back to investigations into imidazole-based sulfonate esters in the late 20th century. Researchers were exploring ways to leverage the nucleophilicity of the imidazole ring to create new functionalized sulfonates. The compound’s unique combination of the aromatic phenyl group, the imidazole ring, and the sulfonate linkage offers chemical stability while retaining reactivity, making it useful for various synthetic applications. The synthesis of phenyl 1H-imidazole-1-sulfonate generally involves the reaction of phenyl sulfonyl chloride with imidazole. This process takes place under mild conditions, often using an organic solvent like dichloromethane or acetonitrile. The imidazole acts as a nucleophile, displacing the chloride group from phenyl sulfonyl chloride to form the sulfonate ester. The reaction typically proceeds smoothly and yields the desired product in high purity after simple purification steps. One of the primary applications of phenyl 1H-imidazole-1-sulfonate is as a reagent for activating hydroxyl groups in organic synthesis. It facilitates the conversion of alcohols into sulfonate esters, which can then undergo substitution reactions to form a wide range of functional groups. This property is particularly useful in the synthesis of pharmaceuticals and bioactive molecules where selective functional group transformations are required. The imidazole group enhances the leaving group ability of the sulfonate, making these transformations efficient and high-yielding. Additionally, phenyl 1H-imidazole-1-sulfonate has found utility in peptide synthesis and other forms of amide bond formation. Its ability to act as a coupling reagent allows chemists to efficiently link amino acids and other building blocks in the construction of peptides and complex organic frameworks. The compound is also employed in the development of new materials, where its structural features contribute to polymer modification and surface functionalization. As research in synthetic chemistry advances, phenyl 1H-imidazole-1-sulfonate continues to be relevant due to its efficiency as a reagent and its adaptability to various synthetic challenges. Its role in facilitating key transformations ensures its place in both academic research and industrial applications. References 2009. Imidazolylsulfonates: Stable and Highly Activated Electrophiles in Cross-Couplings. Synfacts. DOI: 10.1055/s-0029-1216694 2010. Direct C-H Functionalization of Oxazoles Using Sulfamates and Phosphates. Synfacts. DOI: 10.1055/s-0029-1219842 2015. Synthesis of Acyclic and Cyclic Sulfamates: A Review. Synthesis. DOI: 10.1055/s-0034-1378693 |
| Market Analysis Reports |