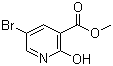
Methyl 5-bromo-2-hydroxynicotinate is an organic compound that consists of a nicotinic acid derivative, where the functional groups include a hydroxyl group (-OH) at the 2-position and a bromine atom at the 5-position of the pyridine ring. Additionally, the compound contains a methyl ester group (-OCH3) attached to the carboxyl group of the nicotinic acid moiety.
This compound is a halogenated derivative of methyl nicotinate, where the presence of the bromine atom introduces unique reactivity and influences the compound's properties. Methyl 5-bromo-2-hydroxynicotinate is of interest in various areas of research due to the presence of both halogen and hydroxyl functionalities, which can provide sites for further chemical modifications, making it a useful intermediate for the synthesis of more complex molecules.
The synthesis of methyl 5-bromo-2-hydroxynicotinate generally involves the selective bromination of methyl 2-hydroxynicotinate at the 5-position. This halogenation reaction can be carried out using brominating agents like bromine or NBS (N-Bromosuccinimide), often under controlled conditions to ensure regioselectivity at the 5-position of the pyridine ring. The hydroxyl group at the 2-position and the ester group at the carboxyl position are typically introduced earlier in the synthesis, and the reaction conditions must be chosen to avoid modifying these groups.
One of the key features of methyl 5-bromo-2-hydroxynicotinate is its potential for further functionalization. The bromine atom can serve as a leaving group in nucleophilic substitution reactions, allowing for the introduction of a wide range of substituents. The hydroxyl group at the 2-position can also participate in hydrogen bonding, influencing the compound's solubility and reactivity. These functional groups make it a useful precursor in the synthesis of more complex organic compounds, including pharmaceuticals, agrochemicals, or materials.
Methyl 5-bromo-2-hydroxynicotinate and related nicotinic acid derivatives have been studied for their potential biological activity. Nicotinic acid and its derivatives are known to interact with various biological targets, including enzymes and receptors, and have been explored for their potential in the treatment of cardiovascular diseases, cancer, and other conditions. The bromine atom in the 5-position may enhance or modulate the compound's bioactivity by altering its electronic properties or interactions with biological targets. The hydroxyl group, on the other hand, may influence the molecule's solubility and ability to cross biological membranes, which is important for its potential as a drug candidate.
In addition to its potential medicinal applications, methyl 5-bromo-2-hydroxynicotinate may also have applications in materials science. Nicotinic acid derivatives are often used in the synthesis of coordination complexes with metal ions. The bromine atom could participate in metal coordination, influencing the properties of the resulting complex. Additionally, the compound could be used in the development of functionalized materials, where the presence of both the hydroxyl group and the bromine atom allows for further tailoring of material properties such as conductivity, stability, or interaction with other molecules.
In conclusion, methyl 5-bromo-2-hydroxynicotinate is a halogenated nicotinic acid derivative with significant potential for use in organic synthesis, medicinal chemistry, and materials science. Its functional groups, including the bromine atom and hydroxyl group, make it a versatile intermediate for further chemical modification and a candidate for bioactive molecule development. Further research into its reactivity and biological properties could lead to novel applications in drug discovery and material development.
|

























 GHS07 Warning Details
GHS07 Warning Details