Online Database of Chemicals from Around the World
| Zhejiang Metallurgical Research Institute Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pgmcatalyst.com | |||
 | +86 (571) 8817-8133 +86 (578) 814-5985 | |||
 | +86 (578) 814-5986 | |||
 | xk@pgmcatalyst.com scm@pgmcatalyst.com ylq@pgmcatalyst.com | |||
| Chemical manufacturer since 1958 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Huzhou Zhuorui Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zrchemtech.com | |||
 | +86 189 6805 0179 | |||
 | sales@zrchemtech.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2020 | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Colonial Metals, Inc. | USA | |||
|---|---|---|---|---|
 | www.colonialmetals.com | |||
 | +1 (410) 398-7200 (800) 962-1537 | |||
 | +1 (410) 398-2918 | |||
 | sales@colonialmetals.com | |||
| Chemical manufacturer | ||||
| HetCat - Heterocycles & Catalysts | Switzerland | |||
|---|---|---|---|---|
 | www.hetcat.com | |||
 | +41 (61) 363-9868 | |||
 | +41 (61) 363-9869 | |||
 | fallahpour@hetcat.com | |||
| Chemical manufacturer | ||||
| MCAT GmbH | Germany | |||
|---|---|---|---|---|
 | www.mcat.de | |||
 | +49 (7531) 939-097 | |||
 | +49 (7531) 939-098 | |||
 | info@mcat.de | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic palladium |
|---|---|
| Name | Allylpalladium chloride dimer |
| Synonyms | Allylpalladium(II) chloride dimer |
| Molecular Structure | 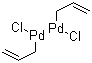 |
| Molecular Formula | C6H10Cl2Pd2 |
| Molecular Weight | 365.89 |
| CAS Registry Number | 12012-95-2 |
| EC Number | 234-579-8 |
| SMILES | [CH2-]C=C.[CH2-]C=C.Cl[Pd+].Cl[Pd+] |
| Melting point | 120 °C (Decomposes) (Expl.) |
|---|---|
| Water solubility | decomposes |
| Hazard Symbols |     GHS02;GHS05;GHS07;GHS09 Danger Details GHS02;GHS05;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H228-H302+H312+H332-H302-H312-H315-H318-H319-H332-H335-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P240-P241-P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P370+P378-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Allylpalladium chloride dimer is an organopalladium compound with the formula [(C3H5)PdCl]2. It consists of two palladium centers, each bonded to an η3-allyl ligand (C3H5) and two bridging chloride ligands connecting the two palladium atoms. This complex is a well-defined, air-stable solid, usually appearing as a yellow to orange crystalline material, and is widely used as a key intermediate and catalyst precursor in organometallic chemistry and homogeneous catalysis. The compound was first reported in the mid-20th century during investigations of transition metal complexes with allyl ligands. Its unique dimeric structure, featuring chlorides bridging two square-planar Pd(II) centers each coordinated by an allyl group, was elucidated through X-ray crystallography and spectroscopic studies. This complex helped to clarify the nature of palladium-allyl bonding and contributed to understanding catalytic cycles involving palladium. Allylpalladium chloride dimer is commonly prepared by the reaction of palladium(II) chloride (PdCl2) with allyl sources such as allyl chloride or allyl acetate under controlled conditions. The reaction typically involves substitution of chloride ligands by the allyl group, resulting in the formation of the dimeric complex with bridging chloride ligands. It can also be prepared by oxidative addition of allyl halides to Pd(0) precursors. In catalysis, allylpalladium chloride dimer serves as a precursor to catalytically active species in numerous palladium-catalyzed transformations. It is widely used in allylic substitution reactions, including Tsuji–Trost reactions, where nucleophiles replace the allyl group coordinated to palladium, enabling the formation of new carbon-carbon or carbon-heteroatom bonds. The complex can be activated by ligands such as phosphines or amines to form monomeric palladium species that participate in catalytic cycles. The dimeric structure provides stability to the palladium complex, facilitating its isolation and handling, but under catalytic conditions the dimer often dissociates to monomeric species. Allylpalladium chloride dimer is soluble in common organic solvents such as dichloromethane, tetrahydrofuran, and acetone, allowing its use in homogeneous catalysis. Apart from allylic substitution, the complex and its derivatives are also involved in cross-coupling reactions, polymerization catalysts, and functionalization of organic molecules. Its reactivity is well-studied and forms the basis of many synthetic methodologies in organic chemistry. From a safety perspective, allylpalladium chloride dimer should be handled with care. Like many palladium compounds, it may cause skin and respiratory irritation, and its dust or solutions should not be inhaled or ingested. It is also a transition metal complex, so environmental disposal must comply with regulations to prevent heavy metal contamination. In summary, allylpalladium chloride dimer is a fundamental palladium(II) complex featuring η3-allyl and bridging chloride ligands. It is widely used as a catalyst precursor in allylic substitution and other palladium-catalyzed transformations, with well-established preparation methods, structural characterization, and applications in synthetic organic chemistry. References none |
| Market Analysis Reports |