Online Database of Chemicals from Around the World
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Sigma-Aldrich, Inc. | China | |||
|---|---|---|---|---|
 | www.sigmaaldrich.com | |||
 | +86 (21) 6141-5566 800-819-3336 | |||
 | orderCN@sial.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Fujian Wolfa Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wolfabio.com | |||
 | +86 18050950397 | |||
 | jomin@wolfabio.com | |||
 | WhatsApp:+86 18359103607 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic scandium, tantalum, thallium, tungsten, antimony, lanthanum, lead, vanadium, molybdenum, chromium, ytterbium, etc. |
|---|---|
| Name | pi-Benzenechromium tricarbonyl |
| Synonyms | eta6-Benzenetricarbonylchromium; chromyl benzene carbon monoxide |
| Molecular Structure | 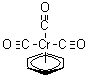 |
| Molecular Formula | C9H6CrO3 |
| Molecular Weight | 214.14 |
| CAS Registry Number | 12082-08-5 |
| EC Number | 235-146-6 |
| SMILES | [Cr].[O+]#[C-].[O+]#[C-].[O+]#[C-].c1ccccc1 |
| Melting point | 163-165 °C* |
|---|---|
| * | Whiting, Mark C. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312+H332 Details | ||||||||||||||||
| Safety Statements | P261-P264-P280-P301+P312-P302+P352+P312-P304+P340+P312 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Pi-benzenechromium tricarbonyl, with the chemical formula Cr(C₆H₆)(CO)₃, is an organometallic compound notable for its unique structure and applications. Also known as chromium hexacarbonyl or chromium(0) tricarbonyl benzene, this compound has had a significant impact on organometallic chemistry and catalysis. The discovery of pi-benzenechromium tricarbonyl can be attributed to advances in organometallic chemistry during the mid-20th century. Researchers exploring transition metal carbonyl complexes discovered that chromium could form stable complexes with carbon monoxide and benzene. The synthesis of this compound involved the reaction of chromium carbonyl with benzene under controlled conditions, resulting in the formation of Cr(C₆H₆)(CO)₃. This discovery was pivotal in expanding the understanding of metal-carbon interactions and led to new insights into the bonding and reactivity of organometallic compounds. One of the primary applications of pi-benzenechromium tricarbonyl is in catalysis. The compound is used as a catalyst in various chemical reactions, particularly in the field of organic synthesis. Its ability to stabilize reactive intermediates and facilitate reactions involving carbon monoxide makes it valuable in processes such as carbonylation and hydroformylation. In carbonylation reactions, pi-benzenechromium tricarbonyl helps introduce carbonyl groups into organic molecules, which is crucial in the production of various chemicals and pharmaceuticals. The compound's role in hydroformylation, where it aids in the addition of formyl groups to alkenes, is similarly important for the synthesis of aldehydes and other chemical intermediates. In addition to its catalytic applications, pi-benzenechromium tricarbonyl has been used in the study of metal-ligand interactions and bonding. The compound serves as a model system for investigating the electronic and geometric effects of transition metal complexes with aromatic ligands. Its well-defined structure and stable coordination make it an ideal candidate for studying the properties of metal-arene interactions and understanding the fundamental principles of organometallic chemistry. The use of pi-benzenechromium tricarbonyl extends to materials science as well. The compound can be employed in the synthesis of chromium-containing materials and coatings with specific properties. For example, it can be used to create chromium-based thin films and nanoparticles with potential applications in electronics and magnetic materials. These materials benefit from the unique properties of chromium and carbonyl groups, contributing to advancements in technology and materials science. Despite its utility, the handling of pi-benzenechromium tricarbonyl requires caution due to the toxicity of carbon monoxide. The compound should be used in well-ventilated areas with appropriate safety measures to avoid exposure to toxic fumes. Proper storage and handling protocols are essential to ensure safe use in laboratory and industrial settings. Ongoing research continues to explore new applications and improve the properties of pi-benzenechromium tricarbonyl. Advances in organometallic chemistry and materials science are leading to the development of novel catalysts and materials based on this compound, with potential implications for various industrial and technological applications. In summary, pi-benzenechromium tricarbonyl is an important organometallic compound with significant applications in catalysis, materials science, and the study of metal-ligand interactions. Its discovery and development have made substantial contributions to the field of chemistry, and its continued research promises to reveal further opportunities for its use in advanced technologies. References 1964. Isotopic exchange of hydrogen atoms of benzenechromium tricarbonyl. Bulletin of the Academy of Sciences of the USSR, Division of chemical science. DOI: 10.1007/bf00849447 2003. Synthesis of ζ6-Fulvene Complexes by Photochemical Substitution of Arenes in (ζ6-Arene)tricarbonylchromium(0). Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-002-00256 2019. Effect of External Electric Field on the Electronic Structure and Aromaticity of Cr(CO)3(ζ6-C6H6) Complex. Russian Journal of Inorganic Chemistry. DOI: 10.1134/s0036023619080114 |
| Market Analysis Reports |