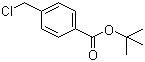
tert-Butyl 4-(chloromethyl)benzoate, with the molecular formula C₁₂H₁₅ClO₂, is an organic compound frequently used as an intermediate in organic synthesis, particularly in the development of pharmaceuticals and agrochemicals. This compound consists of a benzoate ester with a tert-butyl group attached to the carboxyl moiety and a chloromethyl group attached to the benzene ring. The tert-butyl protecting group is notable for its stability under various reaction conditions, while the chloromethyl substituent offers a reactive site for further functionalization. The development of tert-butyl 4-(chloromethyl)benzoate can be traced to advancements in protecting group chemistry during the mid-20th century, when chemists sought stable groups that could withstand diverse synthetic environments yet could be removed without disturbing other sensitive parts of a molecule.
The structure of tert-butyl 4-(chloromethyl)benzoate offers multiple reactive sites, making it a versatile intermediate. The tert-butyl ester is commonly used as a protecting group for carboxylic acids, allowing for selective reactions on other parts of a molecule without affecting the carboxyl group. The chloromethyl group, on the other hand, enables further derivatization by undergoing nucleophilic substitution, alkylation, or coupling reactions. These characteristics have made tert-butyl 4-(chloromethyl)benzoate valuable in medicinal chemistry, where it often serves as a precursor in the synthesis of complex organic molecules, including various pharmaceutical candidates and active compounds.
In applications, tert-butyl 4-(chloromethyl)benzoate is frequently used in the synthesis of molecules that require a protected carboxyl functionality during multi-step synthesis procedures. It is notably applied in the production of aromatic and heteroaromatic compounds, where the chloromethyl group serves as a convenient functional handle for further substitution reactions. The compound’s utility is prominent in the development of drugs and biologically active molecules, where it allows researchers to build complex molecular frameworks in a controlled manner. Additionally, in agrochemical synthesis, tert-butyl 4-(chloromethyl)benzoate acts as a building block for synthesizing herbicides, insecticides, and fungicides, offering a foundation for various substituents required in the active compounds of these agents.
The discovery of tert-butyl 4-(chloromethyl)benzoate has expanded the toolkit for synthetic organic chemists, especially those working on drug discovery and agricultural compounds. Its application in multi-step synthesis has enabled the efficient construction of complex molecules, making it an invaluable intermediate in modern synthetic chemistry.
References
none
|
















 GHS07 Warning Details
GHS07 Warning Details