Online Database of Chemicals from Around the World
| Chizhou Dongsheng Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dspharmchem.com | |||
 | +86 (576) 8429-2571 | |||
 | +86 (576) 8429-2560 | |||
 | ksz@dspharmchem.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Organic raw materials >> Amino compound >> Amide compound |
|---|---|
| Name | 4-tert-Butyl-N-(6-chloro-5-(2-methoxyphenoxy)-2,2'-bipyrimidin-4-yl)benzenesulfonamide |
| Synonyms | N-[6-chloro-5-(2-methoxyphenoxy)[2,2'-bipyrimidin]-4-yl]-4-(1,1-dimethylethyl)benzenesulfonamide |
| Molecular Structure | 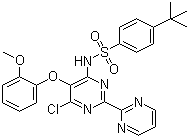 |
| Molecular Formula | C25H26ClN5O4S |
| Molecular Weight | 528.02 |
| CAS Registry Number | 150727-06-3 |
| EC Number | 687-550-0 |
| SMILES | CC(C)(C)C1=CC=C(C=C1)S(=O)(=O)NC2=C(C(=NC(=N2)C3=NC=CC=N3)Cl)OC4=CC=CC=C4OC |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
The chemical substance 4-tert-Butyl-N-(6-chloro-5-(2-methoxyphenoxy)-2,2'-bipyrimidin-4-yl)benzenesulfonamide is a compound primarily noted for its potential in pharmaceutical research, especially within the realm of kinase inhibition. This compound belongs to a class of molecules designed to interfere with signaling pathways that are often dysregulated in diseases such as cancer. The sulfonamide group, combined with the bipyrimidine core, grants the molecule its bioactive properties, allowing it to interact with specific enzymatic targets in a cell. The development of 4-tert-Butyl-N-(6-chloro-5-(2-methoxyphenoxy)-2,2'-bipyrimidin-4-yl)benzenesulfonamide came as part of an effort to explore selective inhibition of protein kinases. These kinases are crucial regulators of cellular functions such as growth, differentiation, and apoptosis. However, in various cancer types, mutations or overexpression of specific kinases lead to uncontrolled cell proliferation. Targeted kinase inhibitors like this compound aim to block these aberrant signaling pathways, potentially stopping the growth of cancer cells while sparing normal cells. Structurally, the compound is characterized by the presence of a bipyrimidine core, a key feature that contributes to its binding affinity for kinase proteins. The methoxyphenoxy group enhances its solubility and bioavailability, while the tert-butyl and sulfonamide groups provide stability and improve its interaction with the kinase active site. These structural attributes enable the compound to interact effectively with the ATP-binding site of kinases, inhibiting their activity. Research into 4-tert-Butyl-N-(6-chloro-5-(2-methoxyphenoxy)-2,2'-bipyrimidin-4-yl)benzenesulfonamide is ongoing, with early studies suggesting its potential in treating certain cancers, particularly those driven by overactive kinases. By selectively inhibiting these kinases, the compound can slow or stop the progression of tumor growth, making it a promising candidate for future cancer therapies. Its bipyrimidine scaffold also makes it a valuable starting point for the development of analogs with improved efficacy and selectivity. Apart from its application in oncology, the compound's structural framework may be applicable in other therapeutic areas where kinase signaling plays a crucial role. For instance, disorders related to inflammation or immune response, such as autoimmune diseases, could potentially benefit from kinase inhibition strategies. However, more research is required to explore these possibilities. As with many kinase inhibitors, there are challenges associated with the use of 4-tert-Butyl-N-(6-chloro-5-(2-methoxyphenoxy)-2,2'-bipyrimidin-4-yl)benzenesulfonamide. Issues such as drug resistance, off-target effects, and toxicity need to be carefully managed in clinical development. Further refinement of the molecule through medicinal chemistry could improve its selectivity and minimize side effects, making it a safer and more effective treatment option. References 2013. An Alternate Synthesis of Bosentan Monohydrate, an Endothelin Receptor Antagonist. Synlett, 24(20). DOI: 10.1055/s-0033-1340281 2003. Bosentan. Pharmaceutical Substances. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-02-0107 |
| Market Analysis Reports |