Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Targetmol China | China | |||
|---|---|---|---|---|
 | www.targetmol.cn | |||
 | +86 (400) 820-0310 | |||
 | sales@targetmol.cn | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Tocris Bioscience Inc. | USA | |||
|---|---|---|---|---|
 | www.tocris.com | |||
 | +1 (636) 207-7651 | |||
 | +1 (636) 207-7683 | |||
 | customerservice.na@bio-techne.com | |||
| Chemical manufacturer since 1982 | ||||
| Classification | API >> Antineoplastic agents |
|---|---|
| Name | Valspodar |
| Synonyms | Cyclo[[(2S,4R,6E)-4-methyl-2-(methylamino)-3-oxo-6-octenoyl]-L-valyl-N-methylglycyl-N-methyl-L-leucyl-L-valyl-N-methyl-L-leucyl-L-alanyl-D-alanyl-N-methyl-L-leucyl-N-methyl-L-leucyl-N-methyl-L-valyl] |
| Molecular Structure | 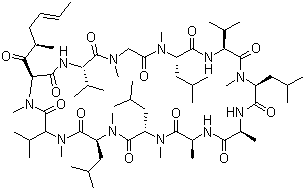 |
| Molecular Formula | C63H111N11O12 |
| Molecular Weight | 1214.62 |
| CAS Registry Number | 121584-18-7 |
| SMILES | C/C=C/C[C@@H](C)C(=O)[C@H]1C(=O)N[C@H](C(=O)N(CC(=O)N([C@H](C(=O)N[C@H](C(=O)N([C@H](C(=O)N[C@H](C(=O)N[C@@H](C(=O)N([C@H](C(=O)N([C@H](C(=O)N([C@H](C(=O)N1C)C(C)C)C)CC(C)C)C)CC(C)C)C)C)C)CC(C)C)C)C(C)C)CC(C)C)C)C)C(C)C |
| Density | 1.0±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 1290.1±65.0 °C 760 mmHg (Calc.)* |
| Flash point | 734.0±34.3 °C (Calc.)* |
| Solubility | 4 Mm in DMSO (Expl.) |
| Index of refraction | 1.467 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| SDS | Available |
|---|---|
|
Valspodar is a synthetic derivative of cyclosporin designed to inhibit P-glycoprotein, a membrane transporter protein involved in the efflux of various drugs from cells. The development of valspodar was motivated by the need to overcome multidrug resistance (MDR) in cancer therapy, where overexpression of P-glycoprotein reduces the intracellular concentration of chemotherapeutic agents, leading to treatment failure. Valspodar was first synthesized and characterized in the 1990s as a non-immunosuppressive analogue of cyclosporin D. Its ability to inhibit P-glycoprotein was demonstrated in various preclinical studies, showing that valspodar can increase the intracellular accumulation of drugs such as doxorubicin, paclitaxel, and vinblastine by blocking their active efflux from resistant cancer cells. This effect made valspodar a promising candidate to enhance the efficacy of chemotherapy in tumors exhibiting multidrug resistance. Clinical development of valspodar focused on its use in combination with conventional chemotherapeutic agents to reverse MDR. Early-phase clinical trials evaluated the safety, pharmacokinetics, and efficacy of valspodar when administered with drugs known to be P-glycoprotein substrates. These studies revealed that valspodar could modulate drug resistance mechanisms and potentially improve therapeutic outcomes in certain cancer types, including hematological malignancies and solid tumors. Despite promising preclinical and initial clinical results, further development faced challenges due to dose-limiting toxicities and complex pharmacokinetic interactions between valspodar and chemotherapeutic agents. These issues limited the maximum tolerated doses of both valspodar and co-administered drugs, complicating the optimization of combination regimens. As a result, valspodar did not achieve widespread clinical approval but remains an important compound in research focused on MDR and P-glycoprotein inhibition. In addition to oncology, valspodar has been used as a research tool to study P-glycoprotein function and drug transport mechanisms in various tissues, including the blood-brain barrier and intestinal epithelium. Its selective inhibition of P-glycoprotein helps elucidate the role of this transporter in pharmacokinetics, drug absorption, and distribution. Overall, valspodar represents a significant advance in the effort to counteract multidrug resistance in cancer therapy through modulation of P-glycoprotein. While clinical use remains limited, valspodar continues to contribute to the understanding of drug transport and resistance mechanisms, informing the design of next-generation inhibitors with improved efficacy and safety profiles. References 1991. Resistance modification by PSC-833, a novel non-immunosuppressive cyclosporin [corrected]. European Journal of Cancer, 27(12). DOI: 10.1016/0277-5379(91)90435-g 1992. Pharmacologic interactions between the resistance‐modifying cyclosporine sdz psc 833 and etoposide (VP 16-213) enhance In Vivo cytostatic activity and toxicity. International Journal of Cancer, 51(3). DOI: 10.1002/ijc.2910510316 1992. SDZ PSC 833 and SDZ 280-446 are the most active of various resistance-modifying agents in restoring rhodamine-123 retention within multidrug resistant P388 cells. Anti-Cancer Drugs, 3(4). DOI: 10.1097/00001813-199208000-00017 |
| Market Analysis Reports |