Online Database of Chemicals from Around the World
| Sichuan Taienkang Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.taienkangpharma.com | |||
 | +86 15102825326 | |||
 | sales@taienkangpharma.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Biochemical >> Amino acids and their derivatives |
|---|---|
| Name | (S)-Methyl 3-amino-3-(4-chlorophenyl)propanoate hydrochloride |
| Synonyms | methyl (3S)-3-amino-3-(4-chlorophenyl)propanoate;hydrochloride |
| Molecular Structure | 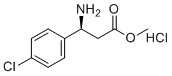 |
| Molecular Formula | C10H13Cl2NO2 |
| Molecular Weight | 250.12 |
| Protein Sequence | X |
| CAS Registry Number | 1217775-76-2 |
| EC Number | 893-521-5 |
| SMILES | COC(=O)C[C@@H](C1=CC=C(C=C1)Cl)N.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
(S)-Methyl 3-amino-3-(4-chlorophenyl)propanoate hydrochloride is a chiral amino acid derivative with significant applications in pharmaceutical synthesis. The discovery of this compound stems from the need to develop enantiomerically pure intermediates for the production of bioactive molecules. Its structure, featuring a chlorophenyl group and an amino acid backbone, is particularly valuable for creating selective and potent therapeutic agents. The synthesis of (S)-Methyl 3-amino-3-(4-chlorophenyl)propanoate hydrochloride involves the asymmetric hydrogenation of α-keto esters using chiral catalysts. This method ensures high enantioselectivity and yield, providing a reliable route to obtain the (S)-enantiomer. The hydrochloride salt form enhances the compound's stability and solubility, facilitating its use in subsequent chemical reactions. This compound plays a crucial role in the development of drugs targeting neurological and cardiovascular disorders. It serves as a key intermediate in the synthesis of inhibitors for enzymes such as proteases and kinases, which are implicated in various disease pathways. Its structural motif is also explored in the design of anticonvulsant and analgesic agents, demonstrating broad pharmaceutical potential. Ongoing research continues to investigate novel applications for (S)-Methyl 3-amino-3-(4-chlorophenyl)propanoate hydrochloride in medicinal chemistry. Its incorporation into drug discovery pipelines highlights the importance of chiral intermediates in modern drug development, underscoring the value of stereochemistry in achieving desired pharmacological effects. References none |
| Market Analysis Reports |