Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Tyger Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.tygersci.com | |||
 | +1 (609) 434-0144 | |||
 | +1 (609) 434-0143 | |||
 | sales@tygersci.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Novasyn Organics Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.novasynorganics.com | |||
 | +91 (406) 620-2233 633-9111 | |||
 | +91 (402) 781-1997 | |||
 | info@novasynorganics.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Pyridine derivative |
|---|---|
| Name | Methyl 3-amino-2-chloroisonicotinate |
| Synonyms | 3-Amino-2-chloroisonicotinic acid methyl ester |
| Molecular Structure | 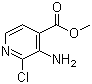 |
| Molecular Formula | C7H7ClN2O2 |
| Molecular Weight | 186.60 |
| CAS Registry Number | 173435-41-1 |
| EC Number | 815-143-1 |
| SMILES | COC(=O)C1=C(C(=NC=C1)Cl)N |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 94 - 96 °C (Expl.) |
| Boiling point | 317.4±37.0 °C 760 mmHg (Calc.)* |
| Flash point | 145.7±26.5 °C (Calc.)* |
| Index of refraction | 1.586 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Methyl 3-amino-2-chloroisonicotinate is a functionalized pyridine derivative that emerged from efforts to explore the reactivity of substituted nicotinic acid esters. Pyridine-based compounds have been foundational in organic chemistry research, particularly for their roles in medicinal and agrochemical applications. Methyl 3-amino-2-chloroisonicotinate was synthesized as part of a broader strategy to introduce multiple reactive groups onto the pyridine ring, thereby enhancing its versatility for subsequent chemical transformations. The compound consists of a pyridine ring bearing a methyl ester group at the 4-position, an amino group at the 3-position, and a chlorine atom at the 2-position. This substitution pattern provides a balance between electron-donating and electron-withdrawing effects, significantly influencing the reactivity of the molecule. The amino group serves as a nucleophilic center, while the chlorine atom can participate in nucleophilic aromatic substitution reactions. These features make Methyl 3-amino-2-chloroisonicotinate a valuable intermediate in the synthesis of more complex heterocyclic systems. In the field of medicinal chemistry, Methyl 3-amino-2-chloroisonicotinate has been used as a key starting material for the development of compounds with antimicrobial, antiviral, and anticancer activities. Its functional groups allow for straightforward derivatization, enabling the construction of a wide array of bioactive molecules. Researchers have utilized it to synthesize a variety of amides, ureas, and carbamates through reactions involving the amino group, while the ester functionality provides opportunities for transesterification or hydrolysis to the corresponding acid, further expanding its utility. The compound's application is not limited to drug development. In agrochemical research, derivatives of Methyl 3-amino-2-chloroisonicotinate have been investigated as potential herbicides and fungicides. The pyridine core structure is known to impart biological activity, and modifications starting from this intermediate have led to the creation of agents that protect crops against a range of pests and diseases. Its amenability to chemical modification facilitates the fine-tuning of biological properties such as potency, selectivity, and environmental stability. Methyl 3-amino-2-chloroisonicotinate is also of interest in the synthesis of advanced materials. Functionalized pyridine derivatives are employed in the design of ligands for metal coordination complexes, which have applications in catalysis and material science. The combination of an amino group and a chlorine substituent allows for tailored interactions with metal centers, making it a candidate for the synthesis of novel coordination compounds with unique electronic or catalytic properties. The practical handling of Methyl 3-amino-2-chloroisonicotinate is relatively straightforward. It is generally stable under ambient conditions and can be stored without significant decomposition. Its solubility in a variety of organic solvents enhances its usability in diverse reaction conditions, including both aqueous and non-aqueous systems. The reactivity of the amino and ester groups, in combination with the potential for displacement of the chlorine atom, supports its role as a versatile building block in synthetic organic chemistry. In synthetic strategies, Methyl 3-amino-2-chloroisonicotinate often serves as an intermediate that allows for divergent synthesis. Starting from this compound, chemists can access a wide range of structures by selectively transforming the amino, ester, or chloro functionalities. This flexibility is especially valuable in drug discovery programs, where the rapid generation of analogs is crucial for structure-activity relationship studies. Overall, Methyl 3-amino-2-chloroisonicotinate is recognized as an important tool in organic synthesis, particularly valued for its capacity to introduce functional diversity into pyridine-based frameworks. Its development and application highlight the ongoing importance of pyridine chemistry in addressing challenges across pharmaceuticals, agriculture, and materials science. |
| Market Analysis Reports |