Online Database of Chemicals from Around the World
| Clearsynth Canada | Canada | |||
|---|---|---|---|---|
 | www.clearsynth.com | |||
 | +1 (415) 685-4395 | |||
 | +1 (289) 729-0064 | |||
 | enquiry@clearsynth.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Chengxi Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.biogrand.com | |||
 | +86 18916572267 | |||
 | chengxitechco@163.com | |||
 | QQ Chat | |||
 | WeChat: +8618916572267 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Amine |
|---|---|
| Name | N-Nitroso-diethylamine D10 |
| Synonyms | N,N-bis(1,1,2,2,2-pentadeuterioethyl)nitrous amide |
| Molecular Structure | 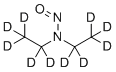 |
| Molecular Formula | C4D10N2O |
| Molecular Weight | 112.20 |
| CAS Registry Number | 1219794-54-3 |
| EC Number | 802-339-7 |
| SMILES | [2H]C([2H])([2H])C([2H])([2H])N(C([2H])([2H])C([2H])([2H])[2H])N=O |
| Density | 0.9±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.442, Calc.* |
| Boiling Point | 173.9±9.0 °C (760 mmHg), Calc.* |
| Flash Point | 59.0±18.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS08 Danger Details GHS06;GHS08 Danger Details |
|---|---|
| Risk Statements | H301-H350 Details |
| Safety Statements | P203-P264-P270-P280-P301+P316-P318-P321-P330-P405-P501 Details |
| SDS | Available |
|
N-Nitroso-diethylamine D10 is a fully deuterated isotopologue of N-nitrosodiethylamine (NDEA), a nitrosamine compound with well-documented carcinogenic properties. The designation D10 indicates that all ten hydrogen atoms in the two ethyl groups of the parent compound are replaced with deuterium (²H), the stable isotope of hydrogen. This isotopic substitution alters the molecular weight without changing the core structure or reactivity of the compound, making it chemically equivalent to the non-labeled version in most analytical applications. N-nitrosodiethylamine itself has been recognized for its toxicological relevance since the mid-20th century, primarily due to its potent hepatocarcinogenic effects observed in laboratory animals. It is part of a broader group of N-nitrosamines, many of which are considered probable human carcinogens. NDEA can be formed as a by-product in various industrial and chemical processes, especially under acidic conditions in the presence of secondary amines and nitrite sources. It has been detected in substances such as tobacco smoke, rubber products, cured meats, and contaminated pharmaceuticals. The deuterated analog, N-nitroso-diethylamine D10, has no therapeutic use but serves a critical function in modern analytical chemistry as an internal standard for mass spectrometry (MS) and gas chromatography-mass spectrometry (GC-MS) techniques. Its primary application is in isotope dilution mass spectrometry, a method that improves the accuracy and precision of quantitative measurements. The mass difference between NDEA and NDEA D10 allows for clear distinction between the analyte and the internal standard during analysis, even in complex sample matrices. This internal standard is especially valuable for detecting trace levels of NDEA in pharmaceutical products, water, food, and biological specimens. Its inclusion in analytical protocols is required by regulatory agencies to ensure rigorous quantification of nitrosamine impurities, which have become a major focus in pharmaceutical quality assurance. Recent regulatory actions by the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other health authorities have established strict permissible limits for N-nitrosamines in drug substances and drug products. In these contexts, N-nitroso-diethylamine D10 enables sensitive detection and validation of analytical methods used to confirm compliance with safety standards. Preparation of this isotopically labeled compound involves the introduction of deuterated ethyl groups through specialized synthetic routes. Commercial suppliers provide the compound in high purity for laboratory use, often accompanied by detailed certificates of analysis that confirm isotopic enrichment and chemical stability. In sum, N-nitroso-diethylamine D10 is a laboratory standard that plays a pivotal role in the detection and quantification of a known carcinogenic impurity. Its reliability and structural similarity to NDEA make it indispensable for analytical laboratories conducting nitrosamine screening and safety assessments across multiple industries. References none |
| Market Analysis Reports |