Online Database of Chemicals from Around the World
| Wuhan Jadechem Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jadechem.com | |||
 | +86 (27) 8355-2071 | |||
 | +86 (27) 8355-1196 | |||
 | info@jadechem-intl.com jhc0005@jadechem.com | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wuhan Organic Synthetic Material Research Institute | China | |||
|---|---|---|---|---|
 | www.whorganic.com | |||
 | +86 (27) 8818-9660 | |||
 | +86 (27) 8341-1474 | |||
 | organic@hi2000.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Jiangsu Mengde New Materials Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinamengde.com | |||
 | +86 (511) 8686-2404 +86 18021229333 | |||
 | +86 (511) 8686-9404 | |||
 | oliverhang@chinamengde.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 1996 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Fengfan International Trade Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.plating-chemistry.com | |||
 | +86 (27) 8561-5818-5 | |||
 | Fcheng@fengfan.net | |||
 | WhatsApp:8613397161982 | |||
| Chemical manufacturer since 1984 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Hubei Sinem Flavor Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinem.cn | |||
 | +86 (27) 8818-9660 | |||
 | sales@sinem.cn | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Classification | Organic raw materials >> Ketone compound |
|---|---|
| Name | Benzalacetone |
| Synonyms | 4-Phenyl-3-buten-2-one; Benzylideneacetone; Methyl styryl ketone |
| Molecular Structure | 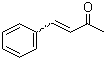 |
| Molecular Formula | C10H10O |
| Molecular Weight | 146.19 |
| CAS Registry Number | 122-57-6 |
| EC Number | 204-555-1 |
| FEMA | 2881 |
| SMILES | CC(=O)/C=C/C1=CC=CC=C1 |
| Density | 1.0±0.1 g/mL, Calc.*, 1.038 g/mL (Expl.) |
|---|---|
| Melting point | 39-42 °C (Expl.) |
| Index of Refraction | 1.563, Calc.* |
| Boiling Point | 260.8±9.0 °C (760 mmHg), Calc.*, 260-262 °C (Expl.) |
| Flash Point | 65.6 °C, Calc.*, 123 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H317-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P272-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Benzalacetone is an organic compound with the chemical formula C9H10O. It is a member of the class of compounds known as α,β-unsaturated ketones, specifically consisting of a benzene ring attached to a propenone group. The compound appears as a pale yellow liquid with a characteristic odor and is used in various industrial and synthetic applications. Benzalacetone was first synthesized in the early 20th century, primarily as a product of the reaction between acetone and benzaldehyde. This reaction, which is a form of aldol condensation, results in the formation of benzalacetone. Its chemical structure, featuring a conjugated system, imparts distinct chemical reactivity, especially in terms of nucleophilic additions and other reactions characteristic of α,β-unsaturated carbonyl compounds. Benzalacetone is most notably used in the flavor and fragrance industry due to its sweet, floral, and somewhat spicy aroma. It is utilized as a flavoring agent in food products, including baked goods, candies, and beverages. Its aromatic properties also make it an important ingredient in perfumes and cosmetics. The compound’s versatility in the fragrance industry is enhanced by its ability to blend well with various other fragrance components, especially in creating floral and fruity notes. In addition to its use in the fragrance and food industries, benzalacetone is employed in the synthesis of other chemicals. It serves as an intermediate in organic chemistry, particularly in the synthesis of heterocyclic compounds. The reactivity of its α,β-unsaturated ketone structure allows for diverse synthetic routes, including the formation of more complex molecules used in pharmaceuticals and agrochemicals. Benzalacetone has also found use in certain polymerization processes and as a component in some resin systems, where it contributes to the properties of the final product. Its reactivity and ability to form derivatives make it valuable in materials science, particularly in the development of specialty coatings and plastics. In terms of safety, benzalacetone is generally regarded as having a low toxicity profile. However, exposure to high concentrations can cause irritation to the skin, eyes, and respiratory system. Proper handling and use of personal protective equipment are recommended during its use in industrial or laboratory settings. In conclusion, benzalacetone is an important chemical used primarily in the fragrance, flavor, and chemical synthesis industries. Its distinctive odor and reactivity make it a useful compound in the production of various consumer products and industrial chemicals. References 1924. Phenylhydräzin als Reagens auf Aldehyde und Ketone. Untersuchungen über Triphenylmethanfarbstoffe Hydrazine und Indole. DOI: 10.1007/978-3-642-51979-6_46 2023. Reactions of Arylaldimines of Glycine Ethyl Ester with Halo-Substituted Arylideneacetones. Russian Journal of General Chemistry. DOI: 10.1134/s1070363223060014 2023. Synthesis of Substituted Cyclohexenones by Reaction of Chalcones with Acetoacetic Acid Amides. Russian Journal of General Chemistry. DOI: 10.1134/s1070363223110026 |
| Market Analysis Reports |