Online Database of Chemicals from Around the World
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | tert-Butyldicyclohexylphosphonium tetrafluoroborate |
| Molecular Structure | 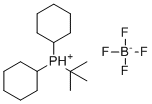 |
| Molecular Formula | C16H32BF4P |
| Molecular Weight | 342.20 |
| CAS Registry Number | 1220349-00-7 |
| SMILES | [B-](F)(F)(F)F.CC(C)(C)[PH+](C1CCCCC1)C2CCCCC2 |
| Melting point | 221-231 °C |
|---|---|
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details |
|---|---|
| Risk Statements | H302-H314 Details |
| Safety Statements | P260-P280-P303+P361+P353-P301+P330+P331-P304+P340+P310-P305+P351+P338+P310 Details |
| SDS | Available |
|
tert-Butyldicyclohexylphosphonium tetrafluoroborate is a specialized organophosphorus compound used primarily in organic synthesis and catalysis. With the formula [(C_6H_11)_2P(C_4H_9)]BF_4, this compound features a tert-butyl group and two cyclohexyl groups bonded to a phosphorus atom, coordinated with a tetrafluoroborate anion. The unique structure of this compound lends it specific properties that make it valuable in various chemical processes. The discovery of tert-butyldicyclohexylphosphonium tetrafluoroborate was part of the broader exploration of phosphonium salts and their role as phase-transfer catalysts and intermediates in organic synthesis. Phosphonium salts, particularly those with bulky substituents, have shown promise in enhancing reaction rates and selectivity by facilitating the transfer of reactants between different phases, such as between organic and aqueous phases. One of the key applications of tert-butyldicyclohexylphosphonium tetrafluoroborate is as a phase-transfer catalyst. Phase-transfer catalysis is a technique used to speed up reactions involving reactants in different phases, typically organic and aqueous phases. The bulky tert-butyl and cyclohexyl groups in this phosphonium salt help to stabilize the catalyst and increase its efficiency in transferring reactants across phase boundaries. This property is particularly useful in reactions such as alkylation and acylation, where the phase-transfer catalyst can significantly enhance the yield and rate of the reaction. In addition to its role in phase-transfer catalysis, tert-butyldicyclohexylphosphonium tetrafluoroborate is also used in the synthesis of various organic compounds. The stability and solubility of this phosphonium salt in different solvents make it a versatile reagent in organic synthesis. It can be employed in reactions where traditional phosphonium salts might be less effective, providing an alternative approach to achieve high yields and selectivity. The tetrafluoroborate anion in the compound contributes to its stability and solubility characteristics. Tetrafluoroborate salts are known for their non-nucleophilic nature, which reduces unwanted side reactions and improves the selectivity of the catalytic processes in which the phosphonium salt is used. This makes tert-butyldicyclohexylphosphonium tetrafluoroborate particularly valuable in delicate synthetic procedures where precise control over reaction conditions is required. Moreover, the use of this phosphonium salt extends to its potential applications in materials science and pharmaceuticals. The ability to precisely control the synthesis of complex molecules and materials using this compound can lead to advancements in developing new materials and drug candidates. In summary, tert-butyldicyclohexylphosphonium tetrafluoroborate is a significant compound in the realm of phase-transfer catalysis and organic synthesis. Its unique structure and properties enable it to act efficiently as a catalyst in various reactions, offering advantages in yield and selectivity. The discovery and application of this phosphonium salt reflect ongoing advancements in catalysis and synthetic chemistry, highlighting its value in both research and industrial applications. References 2012. Effects of Nontransferable Substituent. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-207-00295 2012. Silanolates. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-207-00302 |
| Market Analysis Reports |