Online Database of Chemicals from Around the World
| Shanghai Yeexin Biochem & Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bioyeexin.com | |||
 | +86 (21) 5417-6788 +86 13671761800 | |||
 | +86 (21) 5417-7952 | |||
 | sales@bioyeexin.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Wonderchem Co., Limited | Hong Kong | |||
|---|---|---|---|---|
 | www.wonder-chem.com | |||
 | +852 8192-5218 | |||
 | sales@wonder-chem.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shandong Juntai Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.h2opharm.com | |||
 | +86 15106953682 | |||
 | sales01@h2opharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Biochemical >> Amino acids and their derivatives |
|---|---|
| Name | Fmoc-Aph(Hor)-OH |
| Synonyms | (2S)-3-[4-[[(4S)-2,6-dioxo-1,3-diazinane-4-carbonyl]amino]phenyl]-2-(9H-fluoren-9-ylmethoxycarbonylamino)propanoic acid; 4-({[(4S)-2,6-Dioxohexahydro-4-pyrimidinyl]carbonyl}amino)-N-[(9H-fluoren-9-ylmethoxy)carbonyl]-L-phenylalanine |
| Molecular Structure | 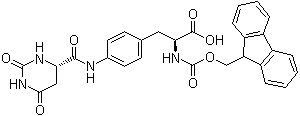 |
| Molecular Formula | C29H26N4O7 |
| Molecular Weight | 542.54 |
| Protein Sequence | X |
| CAS Registry Number | 1253282-31-3 |
| EC Number | 832-774-8 |
| SMILES | C1[C@H](NC(=O)NC1=O)C(=O)NC2=CC=C(C=C2)C[C@@H](C(=O)O)NC(=O)OCC3C4=CC=CC=C4C5=CC=CC=C35 |
| Solubility | Insoluble (1.1E-3 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.412±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.657, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Fmoc-Aph(Hor)-OH, or Fmoc-protected amino acid derivative of 2-amino-3-(4-hydroxyphenyl)propanoic acid, is a chemical compound widely utilized in peptide synthesis and drug development. The discovery of Fmoc-Aph(Hor)-OH emerged from the advancements in solid-phase peptide synthesis (SPPS) in the late 20th century, where protecting group strategies were developed to facilitate the efficient assembly of peptides. The Fmoc (9-fluorenylmethyloxycarbonyl) group serves as a protecting group for the amine functional group of amino acids during peptide synthesis. Its introduction significantly improved the efficiency and ease of the synthesis process, allowing for the selective deprotection of amino acids without affecting other functional groups. The ability to utilize Fmoc-Aph(Hor)-OH in solid-phase synthesis has made it an essential component in the synthesis of peptides containing phenolic residues, enhancing their biological activity and stability. One of the primary applications of Fmoc-Aph(Hor)-OH lies in the synthesis of biologically active peptides and proteins. Its structural characteristics make it a valuable building block for creating peptide analogs with enhanced pharmacological properties. The incorporation of the hydroxyphenyl moiety contributes to the overall hydrophilicity and stability of peptides, which can be beneficial for developing therapeutics targeting various biological pathways. Additionally, Fmoc-Aph(Hor)-OH can be utilized in the design of peptide-based sensors and biomaterials, where its unique properties facilitate specific interactions with target molecules. Fmoc-Aph(Hor)-OH has also been explored for its potential applications in drug delivery systems. The presence of the hydroxyphenyl group may enhance the solubility and stability of peptides in physiological conditions, improving their bioavailability and therapeutic efficacy. Researchers have investigated the use of Fmoc-Aph(Hor)-OH in conjugating peptides to carriers or scaffolds, allowing for targeted delivery to specific cells or tissues. Moreover, Fmoc-Aph(Hor)-OH has garnered interest in the field of medicinal chemistry, where its derivatives are studied for their potential therapeutic effects. The incorporation of the hydroxyphenyl group can modulate the biological activity of peptides, leading to the discovery of novel compounds with improved potency and selectivity for specific targets. Despite its numerous applications, the use of Fmoc-Aph(Hor)-OH in research and industry also necessitates considerations regarding its synthesis, safety, and environmental impact. The development of more sustainable methods for synthesizing and utilizing Fmoc-protected amino acids is an ongoing area of research, aiming to enhance their viability in various applications while minimizing ecological effects. In summary, Fmoc-Aph(Hor)-OH is a significant compound in peptide synthesis, contributing to the development of biologically active peptides, drug delivery systems, and medicinal chemistry. Its discovery and continued exploration highlight its importance as a versatile building block in the advancement of peptide-based therapeutics and biomaterials. References none |
| Market Analysis Reports |