Online Database of Chemicals from Around the World
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Neosun Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neosunpharm.com | |||
 | +86 (21) 2095-6531 +86 18116226605 +86 18939712385 | |||
 | +86 (21) 2095-6532 | |||
 | neosunpharm@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | API >> Nervous system medication |
|---|---|
| Name | Aripiprazole lauroxil |
| Synonyms | Dodecanoic acid [7-[4-[4-(2,3-dichlorophenyl)-1-piperazinyl]butoxy]-3,4-dihydro-2-oxo-1(2H)-quinolinyl]methyl ester; RDC 3317 |
| Molecular Structure | 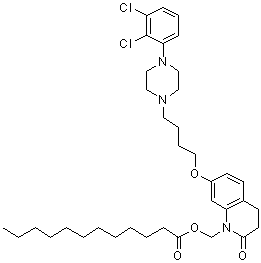 |
| Molecular Formula | C36H51Cl2N3O4 |
| Molecular Weight | 660.71 |
| CAS Registry Number | 1259305-29-7 |
| EC Number | 826-270-7 |
| SMILES | CCCCCCCCCCCC(=O)OCN1C(=O)CCC2=C1C=C(C=C2)OCCCCN3CCN(CC3)C4=C(C(=CC=C4)Cl)Cl |
| Solubility | Insoluble (5.2E-5 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.153±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.551, Calc.* |
| Boiling Point | 781.7±60.0 °C (760 mmHg), Calc.* |
| Flash Point | 426.6±32.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H332-H412 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P317-P321-P330-P362+P364-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Aripiprazole lauroxil is an innovative prodrug formulation of aripiprazole, an atypical antipsychotic used primarily in the treatment of schizophrenia and bipolar disorder. Aripiprazole itself, a partial agonist at dopamine and serotonin receptors, has been utilized for several years to manage symptoms of psychotic disorders. However, its application is often limited by its short half-life, requiring frequent administration. The development of aripiprazole lauroxil was driven by the need for a long-acting injectable formulation that could maintain therapeutic levels of the drug in the body for an extended period, thus improving patient adherence to treatment regimens. The discovery of aripiprazole lauroxil involved the conjugation of aripiprazole with lauroxil, a fatty acid ester that allows for prolonged release. When administered intramuscularly, aripiprazole lauroxil undergoes hydrolysis in the body, releasing the active drug aripiprazole over a period of weeks. This slow release mechanism allows for reduced dosing frequency, improving patient compliance and ensuring more consistent therapeutic levels. The formulation of aripiprazole lauroxil was a significant step forward in addressing the challenges of managing chronic psychiatric conditions, where adherence to oral medications is often problematic. Aripiprazole lauroxil was approved by the U.S. Food and Drug Administration (FDA) in 2015 for the treatment of schizophrenia in adults. Its application extends beyond schizophrenia, as it is also used to treat acute manic and mixed episodes associated with bipolar I disorder. The long-acting nature of the drug makes it particularly beneficial for patients who may have difficulty adhering to daily oral medications, such as those with chronic psychiatric disorders or those in need of stabilization after an acute episode. One of the primary advantages of aripiprazole lauroxil is its ability to maintain therapeutic drug levels over a prolonged period, reducing the need for frequent dosing and minimizing the peaks and troughs in drug concentration that are often seen with oral medications. This leads to more stable treatment outcomes and reduced risk of relapse. Clinical studies have shown that patients receiving aripiprazole lauroxil report fewer symptoms of psychosis and mood instability, along with improved overall functioning compared to those receiving oral aripiprazole. In addition to its use in schizophrenia and bipolar disorder, aripiprazole lauroxil has been explored for potential application in other psychiatric conditions, including major depressive disorder and irritability associated with autism spectrum disorder. Ongoing research aims to expand its indications and optimize dosing regimens to further improve patient care. In conclusion, aripiprazole lauroxil represents a significant advancement in the treatment of psychiatric disorders, offering a more convenient and effective alternative to traditional oral antipsychotic medications. Its extended-release formulation addresses the issue of medication adherence, providing patients with a better chance of sustained symptom management and improved quality of life. References 2018. Population Pharmacokinetic Analysis and Model-Based Simulations of Aripiprazole for a 1-Day Initiation Regimen. Journal of Clinical Pharmacology, 58(9). DOI: 10.1002/jcph.1119 2024. Prolactin Changes Associated with Aripiprazole Lauroxil and Paliperidone Palmitate. Journal of Clinical Psychopharmacology, 44(3). DOI: 10.1097/JCP.0000000000001846 2021. Safety and Efficacy of Aripiprazole Lauroxil in Schizophrenia: A 52-Week Study. Schizophrenia Research, 227. DOI: 10.1016/j.schres.2020.09.013 |
| Market Analysis Reports |