Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Thiophene compound |
|---|---|
| Name | 3,4-Ethylenedioxythiophene |
| Synonyms | 2,3-Dihydrothieno[3,4-b]-1,4-dioxin; EDOT |
| Molecular Structure | 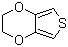 |
| Molecular Formula | C6H6O2S |
| Molecular Weight | 142.17 |
| CAS Registry Number | 126213-50-1 |
| EC Number | 415-450-7 |
| SMILES | C1COC2=CSC=C2O1 |
| Density | 1.331 g/mL (Expl.) |
|---|---|
| Boiling point | 193 °C (Expl.) |
| Refractive index | 1.5765 (Expl.) |
| Flash point | 104 °C (Expl.) |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H311-H312-H319-H412 Details | ||||||||||||||||||||||||||||
| Safety Statements | P262-P264-P264+P265-P270-P273-P280-P301+P317-P302+P352-P305+P351+P338-P316-P317-P321-P330-P337+P317-P361+P364-P362+P364-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 2810 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
3,4-Ethylenedioxythiophene (EDOT) is an organic compound of significant interest in materials science and electronic engineering. Its structure, a thiophene ring fused with an ethylenedioxy group, gives it unique electronic and chemical properties that have driven its discovery and widespread application. EDOT was first synthesized in the late 20th century as part of efforts to develop novel conductive monomers for use in advanced polymer systems. The discovery of EDOT stemmed from the exploration of thiophene derivatives for their potential in creating conductive polymers. Researchers identified that the ethylenedioxy group enhanced the electron-donating properties of the thiophene ring, facilitating the formation of conductive polymers with improved stability and conductivity. This breakthrough laid the foundation for the development of poly(3,4-ethylenedioxythiophene) (PEDOT), a material that remains central to modern electronics. EDOT polymerizes readily, producing PEDOT, which is valued for its high electrical conductivity, optical transparency in thin films, and environmental stability. These properties make PEDOT an essential material in applications such as organic light-emitting diodes (OLEDs), solar cells, and antistatic coatings. The development of PEDOT:PSS, a water-dispersible composite of PEDOT and polystyrene sulfonate, further expanded its usability, allowing for straightforward processing in flexible electronic devices and sensors. Beyond electronics, EDOT has found uses in energy storage systems, such as supercapacitors and batteries, where its polymer exhibits excellent charge transport and cycling stability. It is also utilized in biomedical applications, including biosensors and neural interfaces, due to its biocompatibility and ability to facilitate electrical signal transmission in biological environments. The discovery and development of EDOT underscore the importance of small-molecule innovations in advancing material technologies. Its applications across diverse fields highlight its versatility and the enduring impact of conductive polymers on modern science and industry. References 1996. Electrochromic Properties of PEDOT. Advanced Materials, 8(10). DOI: 10.1002/adma.19960081003 2024. Electrodeposited Composite of Poly-3,4-ethylenedioxythiophene with Fullerenol Photoactive in the Near-IR Range. Journal of Physical Chemistry C, 128(12). DOI: 10.1021/acs.jpcc.4c00234 2024. PEDOT-Based Composites for Electrochemical Sensors. Journal of Materials Chemistry C, 12(15). DOI: 10.1039/D4TC00123A |
| Market Analysis Reports |