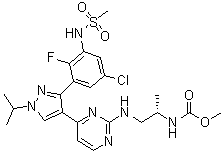
Encorafenib is a potent and selective inhibitor of BRAF kinase, which plays a critical role in the mitogen-activated protein kinase (MAPK) pathway. This pathway is often implicated in the development of various cancers, including melanoma. Encorafenib was developed to overcome limitations observed with earlier BRAF inhibitors, such as resistance and limited efficacy. The discovery of Encorafenib marked a significant advancement in targeted cancer therapies by improving selectivity and reducing off-target effects.
The synthesis of Encorafenib involves a complex multi-step process designed to optimize yield and purity. Key steps in the synthesis include the formation of the pyridine and arylamine core structures, which are critical for its activity. Several research groups have refined synthetic strategies to ensure scalability and reproducibility, essential for clinical and commercial production. Encorafenib's synthesis typically employs palladium-catalyzed coupling reactions and selective fluorination techniques, which contribute to the compound's stability and pharmacokinetic properties.
Encorafenib has been extensively studied for its role in treating BRAF V600E-mutant metastatic melanoma, often in combination with MEK inhibitors such as binimetinib. Clinical trials have demonstrated that this combination significantly improves progression-free survival compared to BRAF inhibitor monotherapy. Encorafenib's application extends beyond melanoma, with ongoing investigations exploring its efficacy in colorectal cancer and other malignancies driven by BRAF mutations. The ability to inhibit tumor growth through precise targeting has made Encorafenib a cornerstone in precision oncology.
References
2012. The role of BRAF V600 mutation in melanoma. Journal of Translational Medicine, 10.
DOI: 10.1186/1479-5876-10-85
2024. Overcoming Resistance Mechanisms to Melanoma Immunotherapy. American Journal of Clinical Dermatology, 25(6).
DOI: 10.1007/s40257-024-00907-7
2024. Safe Integration of Encorafenib plus Binimetinib with Stereotactic Radiosurgery for Melanoma Brain Metastases: a Case Report. La Clinica terapeutica, 175(6).
DOI: 10.7417/ct.2024.5141
|






















 GHS08 Danger Details
GHS08 Danger Details