Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2010 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Isoquinoline compound |
|---|---|
| Name | 5,7-Dichloro-1,2,3,4-tetrahydro-6-isoquinolinecarboxylic acid hydrochloride (1:1) |
| Molecular Structure | 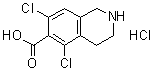 |
| Molecular Formula | C10H9Cl2NO2.HCl |
| Molecular Weight | 282.55 |
| CAS Registry Number | 1289646-93-0 |
| EC Number | 815-386-3 |
| SMILES | C1CNCC2=CC(=C(C(=C21)Cl)C(=O)O)Cl.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312+H332-H302-H312-H315-H319-H332-H335-H412 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
5,7-Dichloro-1,2,3,4-tetrahydro-6-isoquinolinecarboxylic acid hydrochloride (1:1) is a chemical compound that features an isoquinoline core structure with chlorination at positions 5 and 7. This compound belongs to a broader class of isoquinoline derivatives, which are of considerable interest in medicinal chemistry due to their bioactive properties. Isoquinolines, as heterocyclic compounds, possess a wide range of pharmacological activities, including antimicrobial, anti-inflammatory, and anticancer properties. The incorporation of chlorine atoms into the isoquinoline structure has been shown to modulate its biological activity, making compounds like 5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinecarboxylic acid hydrochloride promising candidates for drug development. The discovery of 5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinecarboxylic acid hydrochloride was driven by the desire to develop compounds that could target specific enzymes and receptors in the body. The tetrahydroisoquinoline ring system is known for its ability to bind to various biological targets, including enzymes involved in neurotransmitter metabolism and receptors that modulate cellular signaling. The addition of chloro groups to the isoquinoline ring likely enhances its interaction with these targets, improving the compound's potency and selectivity. Moreover, the carboxylic acid functional group provides a point of interaction with biological macromolecules, potentially increasing its solubility and pharmacokinetic properties. The application of 5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinecarboxylic acid hydrochloride has been explored in the development of treatments for a variety of diseases. Research suggests that this compound could be useful in the treatment of neurological disorders, as it may act on specific receptors involved in the regulation of neurotransmitter levels. Additionally, due to its structural similarity to other bioactive isoquinoline derivatives, it has potential as an anticancer or antimicrobial agent. The compound's ability to modulate cellular pathways related to inflammation and immune response also makes it a candidate for further investigation in the development of anti-inflammatory therapies. In pharmaceutical applications, 5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinecarboxylic acid hydrochloride is considered an important building block for the synthesis of more complex molecules. The ability to modify the structure by introducing different functional groups allows researchers to optimize the compound's efficacy and specificity. As such, it serves as a key intermediate in the synthesis of drugs targeting a wide array of diseases, including cancer, neurodegenerative disorders, and infections. The ongoing research into 5,7-dichloro-1,2,3,4-tetrahydro-6-isoquinolinecarboxylic acid hydrochloride highlights its potential as a versatile pharmacological agent. Future studies will continue to explore its molecular interactions, biological effects, and possible therapeutic applications. References none |
| Market Analysis Reports |