Online Database of Chemicals from Around the World
| MolScanner | Singapore | |||
|---|---|---|---|---|
 | www.molscanner.com | |||
 | +86 18621675448 | |||
 | marketing@molscanner.com | |||
 | WhatsApp:9896 7603 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Organic fluorine compound >> Fluorobenzoic acid series |
|---|---|
| Name | 3-Fluoro-2-(pyrimidin-2-yl)benzoic acid |
| Molecular Structure | 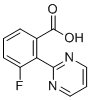 |
| Molecular Formula | C11H7FN2O2 |
| Molecular Weight | 218.18 |
| CAS Registry Number | 1293285-04-7 |
| SMILES | C1=CC(=C(C(=C1)F)C2=NC=CC=N2)C(=O)O |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 304.8±42.0 °C 760 mmHg (Calc.)* |
| Flash point | 138.2±27.9 °C (Calc.)* |
| Index of refraction | 1.599 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H312-H332 Details |
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P330-P363-P501 Details |
| SDS | Available |
|
3-Fluoro-2-(pyrimidin-2-yl)benzoic acid is an aromatic compound consisting of a benzoic acid core substituted at positions 2 and 3 with a pyrimidine ring and a fluorine atom, respectively. This structure features a fluorinated benzoic acid derivative with a pyrimidine group, which imparts distinctive chemical properties to the compound. The **benzoic acid** part of the molecule provides a carboxylic acid group (-COOH), which is a common functional group in organic synthesis and medicinal chemistry. The carboxyl group can participate in a variety of reactions, including esterification, amide formation, and decarboxylation. It is also a hydrophilic group, which contributes to the solubility of the molecule in water and polar solvents. The **fluorine atom** at position 3 of the benzoic acid ring enhances the electronic properties of the molecule. Fluorine is a highly electronegative element, and its presence in aromatic compounds often increases the molecule's stability by inductively withdrawing electron density from the ring. This electron-withdrawing effect can affect the reactivity of the compound, particularly in electrophilic substitution reactions. The fluorine substitution may also influence the compound's pharmacological properties, such as bioavailability or receptor binding affinity, by altering its interaction with biological targets. The **pyrimidine ring** at position 2 of the benzoic acid core is a six-membered heterocyclic structure containing two nitrogen atoms at positions 1 and 3. Pyrimidine derivatives are important in medicinal chemistry due to their presence in many bioactive molecules, including drugs and nucleic acids. The nitrogen atoms in the pyrimidine ring can participate in hydrogen bonding interactions with biological macromolecules, such as enzymes or receptors, enhancing the binding affinity and specificity of the compound. Pyrimidine-containing compounds are often used in the design of antiviral, anticancer, and antimicrobial agents. In terms of **applications**, 3-fluoro-2-(pyrimidin-2-yl)benzoic acid could be explored for its potential bioactivity. The pyrimidine and benzoic acid motifs are commonly found in compounds with antimicrobial, anticancer, or enzyme-inhibiting properties. The fluorine atom may further modulate the compound’s properties, such as increasing its stability or optimizing its interaction with specific targets in the body. The **synthesis** of this compound could be achieved through standard functionalization of the benzoic acid ring and subsequent coupling with the pyrimidine ring. One potential synthetic route involves the introduction of the fluorine atom via electrophilic fluorination of the benzoic acid precursor, followed by the addition of the pyrimidine group, possibly through a nucleophilic aromatic substitution reaction or via cross-coupling methods. In summary, 3-fluoro-2-(pyrimidin-2-yl)benzoic acid is a compound with potential applications in medicinal chemistry, particularly in the development of drugs targeting nucleic acid-related processes or enzymes. The combination of the fluorine atom, carboxylic acid group, and pyrimidine ring provides a structurally interesting molecule that could have enhanced stability and specific interactions with biological targets. Its design and reactivity suggest potential use in various therapeutic areas, including cancer and infection treatment. |
| Market Analysis Reports |