1,4-Naphthoquinone, a member of the naphthoquinone family, is a bicyclic organic compound with the molecular formula C₁₀H₆O₂. It features a quinone functional group and has gained significant attention due to its diverse applications across various fields, including organic chemistry, biochemistry, and materials science.
The discovery of 1,4-naphthoquinone dates back to the early 19th century. It was first isolated from the oxidation of naphthalene, a polycyclic aromatic hydrocarbon commonly derived from coal tar. Researchers identified this compound through its distinctive yellow color and its characteristic reactivity. Subsequent studies revealed its potential for further transformation into other valuable derivatives, contributing to its popularity in synthetic chemistry.
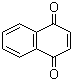
In terms of structure, 1,4-naphthoquinone consists of two fused aromatic rings, with two carbonyl groups located at the 1 and 4 positions of the naphthalene framework. This arrangement imparts unique electronic and chemical properties, enabling it to participate in various chemical reactions, such as electrophilic substitutions, reductions, and complexation with metal ions.
One of the most notable applications of 1,4-naphthoquinone is in the synthesis of dyes and pigments. Its intense color and ability to form complexes with metals make it an essential ingredient in the production of textile dyes and printing inks. Additionally, 1,4-naphthoquinone is employed as a precursor for the synthesis of other valuable compounds, including pharmaceuticals and agrochemicals. For instance, it serves as a building block for the synthesis of anticoagulants, anti-inflammatory agents, and herbicides, highlighting its importance in medicinal chemistry.
Furthermore, 1,4-naphthoquinone exhibits biological activity, which has spurred research into its potential therapeutic applications. Studies have shown that it possesses antimicrobial and antifungal properties, making it a candidate for the development of new antimicrobial agents. Its ability to induce oxidative stress in cells has also led to investigations into its role in cancer research, where it may serve as a tool for understanding cancer cell metabolism and viability.
In materials science, 1,4-naphthoquinone is used as a photoresist in photolithography processes, particularly in the fabrication of electronic components. Its photo-reactive properties allow for the precise patterning of materials, which is crucial in microelectronics. Additionally, it has been explored as a potential dopant in organic electronics, contributing to advancements in organic light-emitting diodes (OLEDs) and organic photovoltaics (OPVs).
Overall, 1,4-naphthoquinone stands out as a versatile chemical compound with a rich history of discovery and a broad spectrum of applications. Its unique structural features enable it to participate in various chemical reactions, making it an invaluable resource in synthetic chemistry, biochemistry, and materials science. Ongoing research continues to unveil new potentials and applications for this compound, underscoring its significance in modern chemistry.
References
1984. Metabolic activation of 1-naphthol by rat liver microsomes to 1,4-naphthoquinone and covalent binding species. Biochemical Pharmacology, 33(20).
DOI: 10.1016/0006-2952(84)90077-7
2012. Reviews on 1,4-naphthoquinones from Diospyros L. Journal of Asian Natural Products Research, 14(1).
DOI: 10.1080/10286020.2011.633515
2012. First synthesis and anticancer activity of novel naphthoquinone amides. European Journal of Medicinal Chemistry, 49.
DOI: 10.1016/j.ejmech.2012.01.020
|





















































 GHS05;GHS06;GHS07;GHS08;GHS09 Danger Details
GHS05;GHS06;GHS07;GHS08;GHS09 Danger Details