Online Database of Chemicals from Around the World
| CAS: 130137-05-2 Product: 4,5-Difluoro-2-iodobenzoic acid No suppliers available. |
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | 4,5-Difluoro-2-iodobenzoic acid |
| Molecular Structure | 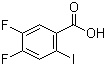 |
| Molecular Formula | C7H3F2IO2 |
| Molecular Weight | 284.00 |
| CAS Registry Number | 130137-05-2 |
| EC Number | 630-211-9 |
| SMILES | C1=C(C(=CC(=C1F)F)I)C(=O)O |
| Boiling point | 305.7±42.0 °C 760 mmHg (Calc.)* |
|---|---|
| Flash point | 138.7±27.9 °C (Calc.)* |
| Index of refraction | 1.613 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
4,5-Difluoro-2-iodobenzoic acid is an organic compound that consists of a benzene ring with three substituent groups: two fluorine atoms at the 4- and 5-positions and an iodine atom at the 2-position. The compound also contains a carboxylic acid group (-COOH) attached at the 1-position of the benzene ring. This structure places the molecule in the class of halogenated benzoic acids, which have applications in both organic synthesis and materials science. The synthesis of 4,5-difluoro-2-iodobenzoic acid generally involves the selective introduction of the iodine and fluorine atoms into the aromatic ring. Typically, the iodo group is introduced first via iodination reactions, often using iodine in the presence of a suitable oxidizing agent or using an iodine source such as N-iodosuccinimide (NIS). The fluorine atoms can be introduced through fluorination reactions, which may involve electrophilic fluorination with reagents like N-fluorobenzenesulfonimide (NFSI) or by using fluorinating agents such as potassium fluoride (KF) under controlled conditions to direct the fluorine atoms to the desired positions on the ring. 4,5-Difluoro-2-iodobenzoic acid is of interest for its potential utility in medicinal chemistry, particularly in the design and synthesis of bioactive compounds. The fluorine and iodine substituents can influence the electronic properties of the aromatic ring, making it more reactive toward certain types of reactions. Fluorine, in particular, can enhance the metabolic stability of a compound by making it more resistant to enzymatic degradation, while iodine atoms can introduce opportunities for further modification via nucleophilic substitution or coordination with metal centers. In medicinal chemistry, halogenated benzoic acids like 4,5-difluoro-2-iodobenzoic acid are studied for their potential biological activity. The halogen atoms can modulate the binding properties of the molecule to various receptors, enzymes, or ion channels, making such compounds candidates for further testing in the development of pharmaceuticals. For example, compounds with similar structures have been explored for their ability to inhibit specific enzymes or as part of more complex scaffolds in the search for anti-inflammatory, anticancer, or antimicrobial agents. In addition to its medicinal potential, 4,5-difluoro-2-iodobenzoic acid may also have applications in materials science. The functional groups on the benzene ring—particularly the carboxylic acid group—can participate in coordination chemistry, making the compound useful as a precursor for the synthesis of metal-organic frameworks (MOFs) or other coordination complexes. The presence of halogen atoms can influence the properties of these complexes, such as their stability, solubility, or conductivity. Furthermore, the compound could be used in the development of advanced materials for sensors, electronic devices, or catalysts. The incorporation of both fluorine and iodine atoms on the same aromatic ring can also affect the physical properties of the compound, such as its solubility, melting point, and spectral characteristics. These modifications make 4,5-difluoro-2-iodobenzoic acid an interesting candidate for use in organic electronics, including in the fabrication of organic light-emitting diodes (OLEDs) or other optoelectronic devices. The fluorine atoms, in particular, may provide stability against oxidation or degradation in such applications. In conclusion, 4,5-difluoro-2-iodobenzoic acid is a halogenated aromatic compound that exhibits a range of potential applications in both medicinal chemistry and materials science. Its unique structure, containing both fluorine and iodine atoms, offers opportunities for further functionalization, making it a versatile intermediate in organic synthesis. Further exploration of its biological and physical properties could lead to the development of new therapeutic agents or advanced materials with specialized applications. |
| Market Analysis Reports |