Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Changzhou Jiuwu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuwuchem.com | |||
 | +86 (519) 8512-1086 +86 13775162768 | |||
 | +86 (519) 8512-5369 | |||
 | info@jiuwuchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Jiangxi Fangzun Pharmaceutical & Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fangzunchem.com | |||
 | +86 15961115886 | |||
 | sales@baokangchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| Achemica | Switzerland | |||
|---|---|---|---|---|
 | www.achemica.com | |||
 | +41 (24) 466-2929 | |||
 | +41 (24) 466-2800 | |||
 | contact@achemica.com | |||
| Chemical manufacturer since 2010 | ||||
| SynInnova Laboratories Inc. | USA | |||
|---|---|---|---|---|
 | www.syninnova.com | |||
 | +1 (780) 485-2962 | |||
 | +1 (780) 485-2963 | |||
 | sales@syninnova.com | |||
| Chemical manufacturer | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Methyl 2-bromo-3-methylbenzoate |
| Molecular Structure | 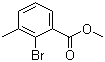 |
| Molecular Formula | C9H9BrO2 |
| Molecular Weight | 229.07 |
| CAS Registry Number | 131001-86-0 |
| EC Number | 811-115-8 |
| SMILES | CC1=C(C(=CC=C1)C(=O)OC)Br |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.545, Calc.* |
| Boiling Point | 267.4±20.0 °C (760 mmHg), Calc.* |
| Flash Point | 115.5±21.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Methyl 2-bromo-3-methylbenzoate is an aromatic ester widely recognized for its importance in organic synthesis. Its structure, featuring a bromine atom and a methyl group attached to the benzene ring, as well as a methyl ester functional group, gives the compound unique reactivity and versatility, making it a valuable intermediate in pharmaceuticals, agrochemicals, and materials science. The discovery of methyl 2-bromo-3-methylbenzoate stems from the development of halogenated aromatic compounds, which have been extensively studied for their utility in functional group transformations. The synthesis typically involves the bromination of 3-methylbenzoic acid derivatives, followed by esterification. Bromination is commonly performed using brominating agents such as bromine or N-bromosuccinimide (NBS), while the esterification step often employs methanol and acid catalysts to yield the methyl ester. In the pharmaceutical industry, methyl 2-bromo-3-methylbenzoate is used as a precursor for synthesizing various bioactive compounds. The bromine atom serves as a handle for further functionalization, enabling the introduction of complex groups through palladium-catalyzed cross-coupling reactions, such as Suzuki, Heck, or Sonogashira couplings. These transformations facilitate the creation of molecules with potential anti-inflammatory, antimicrobial, or anticancer activities. In agrochemical research, methyl 2-bromo-3-methylbenzoate is instrumental in developing pesticides and herbicides. Its aromatic framework and reactive bromine site allow the design of compounds that effectively target pests or weeds while minimizing environmental impact. Researchers have utilized this compound in structure-activity relationship (SAR) studies to optimize the performance of agrochemicals. The compound also finds applications in materials science, particularly in the synthesis of advanced polymers and organic electronics. Its bromine functionality enables incorporation into complex polymer backbones, enhancing thermal stability and electronic properties. Additionally, it serves as a building block for dyes and pigments with applications in industrial coatings and printing technologies. Ongoing research is focused on expanding the scope of reactions using methyl 2-bromo-3-methylbenzoate as a key intermediate. For example, the development of sustainable and regioselective functionalization methods aims to improve synthetic efficiency and reduce waste. Moreover, its inclusion in combinatorial libraries allows for high-throughput screening of potential new materials and bioactive molecules. References 2020. Suzuki-Miyaura Cross Coupling of (2-Hydroxyaryl)boronic Acids with 2-Bromobenzoates. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-114-00535 2020. Palladium-Catalyzed Decarboxylative Cross Coupling and Lactonization. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-114-00537 2024. Ni-catalysed assembly of axially chiral alkenes from alkynyl tetracoordinate borons via 1,3-metallate shift. Nature Chemistry, 16(1). DOI: 10.1038/s41557-023-01396-7 |
| Market Analysis Reports |