Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| PharmFar Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharmfar.com | |||
 | +86 (573) 8751-4118 | |||
 | +86 (573) 8359-1319 | |||
 | sales@pharmfar.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Tianjin Collabrate Chemical Industry. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemcollabrate.com | |||
 | +86 (22) 6330-1939 | |||
 | +86 (22) 6315-2797 | |||
 | catherine_liu@chemcollabrate.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Xuchang Chenhe Bio-pharmaceutical Tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.the-chem.com | |||
 | +86 (374) 579-3128 +86 15836530076 | |||
 | +86 (374) 579-3128 | |||
 | wxh5168@126.com wxh5168@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Bosche Scientific LLC, | USA | |||
|---|---|---|---|---|
 | www.boschesci.com | |||
 | +1 (732) 565-9988 | |||
 | +1 (732) 875-0899 | |||
 | sales@boschesci.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Accel Pharmtech, LLC | USA | |||
|---|---|---|---|---|
 | www.accelpharmtech.com | |||
 | +1 (732) 993-9004 | |||
 | +1 (732) 289-6244 | |||
 | info@accelpharmtech.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Thiophene compound |
|---|---|
| Name | S-(+)-N,N-Dimethyl-3-(1-naphthoxy)-3-(2-thienyl)-1-propylamine oxalate |
| Synonyms | S-(+)-N,N-Dimethyl-3-(1-naphthalenyloxy)-3-(2-thienyl)-1-propanamine oxalate; (S)-N,N-Dimethyl-g-(1-naphthalenyloxy)-2-thiophenepropanamine ethanedioate (1:1) |
| Molecular Structure | 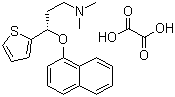 |
| Molecular Formula | C19H21NOS.C2H2O4 |
| Molecular Weight | 401.48 |
| CAS Registry Number | 132335-47-8 |
| EC Number | 603-567-8 |
| SMILES | CN(C)CC[C@@H](C1=CC=CS1)OC2=CC=CC3=CC=CC=C32.C(=O)(C(=O)O)O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
S-(+)-N,N-Dimethyl-3-(1-naphthoxy)-3-(2-thienyl)-1-propylamine oxalate, often abbreviated as S-(+)-NAP, was discovered through rigorous chemical synthesis and screening processes. Developed by pharmaceutical researchers, its synthesis involved intricate steps to achieve the desired stereochemistry, resulting in a potent and selective compound. S-(+)-NAP exhibits promising potential in the treatment of neurological disorders, particularly Parkinson's disease. It acts as a selective agonist of dopamine receptors, effectively modulating dopamine levels in the brain. This mechanism offers neuroprotective effects, potentially slowing down the progression of Parkinson's and mitigating its symptoms. Researchers are exploring the antidepressant properties of S-(+)-NAP due to its ability to enhance serotonin and norepinephrine neurotransmission. By regulating the levels of these neurotransmitters, S-(+)-NAP may offer a novel approach to managing depression and related mood disorders. Preliminary studies suggest that S-(+)-NAP could aid in the management of drug addiction by targeting the brain's reward system. Its action on specific neurotransmitter pathways holds promise in reducing cravings and withdrawal symptoms associated with substance abuse disorders. Neuropathic pain, a debilitating condition resulting from nerve damage, poses significant therapeutic challenges. S-(+)-NAP's modulation of neurotransmitter activity offers a potential avenue for alleviating neuropathic pain, improving patients' quality of life. Traumatic brain injury often leads to long-term neurological deficits due to secondary injury mechanisms. S-(+)-NAP's neuroprotective properties, including anti-inflammatory and antioxidant effects, present a promising strategy for mitigating damage following TBI and promoting recovery. Early research indicates that S-(+)-NAP may hold therapeutic potential in Alzheimer's disease by targeting amyloid-beta aggregation and neuroinflammation, key pathological processes in the disease's progression. References none |
| Market Analysis Reports |