Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Boyuan Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.boyuanpharm.com | |||
 | +86 (531) 6995-4981 8896-3280 +86 15806417970 | |||
 | +86 (531) 8896-4879 | |||
 | Jeffrey.Liu@boyuanpharm.com boyuanchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives |
|---|---|
| Name | 1,2-Cyclohexylenedinitrilotetraacetic acid |
| Synonyms | CDTA; CyDTA; trans-1,2-Diaminocyclohexane-N,N,N',N'-tetraacetic acid; trans-1,2-Cyclohexanediamine-N,N,N',N'-tetraacetic acid |
| Molecular Structure | 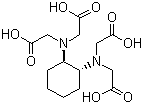 |
| Molecular Formula | C14H22N2O8 |
| Molecular Weight | 346.34 |
| CAS Registry Number | 13291-61-7 |
| EC Number | 236-308-9 |
| SMILES | C1CC[C@H]([C@@H](C1)N(CC(=O)O)CC(=O)O)N(CC(=O)O)CC(=O)O |
| Density | 1.5$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 213-216 $degree$C (Expl.) |
| Boiling point | 670.8$+/-$55.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 359.5$+/-$31.5 $degree$C (Calc.)* |
| Index of refraction | 1.596 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
1,2-Cyclohexylenedinitrilotetraacetic acid is a polyaminocarboxylic acid chelating agent containing a cyclohexane backbone substituted with two nitrogen atoms, each bearing acetic acid groups. It is commonly abbreviated as CDTA and belongs to the same general class of aminopolycarboxylate chelators as ethylenediaminetetraacetic acid (EDTA). The compound is known for its strong ability to bind metal ions through multidentate coordination. Structurally, the molecule consists of a cyclohexane ring with adjacent carbon atoms substituted by nitrogen-containing acetate arms. Each nitrogen atom carries two acetic acid substituents, resulting in a total of four carboxylic acid groups and two tertiary amine donor sites. This arrangement creates a hexadentate ligand capable of coordinating strongly to a wide range of metal cations. The cyclohexane backbone distinguishes CDTA from EDTA, which contains a more flexible ethylene bridge between nitrogen atoms. The rigid cyclic structure of CDTA influences the spatial orientation of donor atoms and often results in stronger and more selective metal complexation for certain ions. The stereochemistry of the cyclohexane ring also affects complex stability, as different conformations can alter donor atom geometry. Chelation occurs through donation of electron pairs from the nitrogen atoms and deprotonated carboxylate oxygen atoms to a central metal ion. In aqueous solution, the carboxylic acid groups can lose protons to form negatively charged carboxylates, which strongly coordinate positively charged metal ions. This multidentate binding produces highly stable chelate complexes. 1,2-Cyclohexylenedinitrilotetraacetic acid has been widely studied in coordination chemistry and analytical chemistry because of its high affinity for divalent and trivalent metal ions. It forms especially stable complexes with alkaline earth metals and transition metals, including calcium, magnesium, copper, iron, and rare earth elements. The enhanced complex stability relative to EDTA for some metals has made CDTA useful in specialized titration and separation procedures. In analytical chemistry, CDTA is used as a complexometric titration reagent and masking agent. By selectively binding certain metal ions, it allows accurate quantitative determination of metals in mixtures. The stability constants of CDTA complexes are often higher than those of analogous EDTA complexes, particularly for calcium and other larger metal ions. The compound also has applications in industrial and laboratory processes involving metal ion sequestration. Chelating agents such as CDTA are used to prevent unwanted precipitation, reduce metal-catalyzed oxidation reactions, and control trace metal contamination in chemical systems. Synthesis of CDTA generally involves reaction of trans-1,2-diaminocyclohexane with chloroacetic acid or related haloacetic acid derivatives under alkaline conditions. The nitrogen atoms undergo alkylation to introduce the acetate arms, producing the tetraacetic acid derivative after hydrolysis and acidification. From a physicochemical standpoint, CDTA is highly polar and capable of forming multiple ionic species depending on pH. The acid–base behavior of its four carboxylic acid groups strongly influences metal-binding properties. At higher pH, deprotonation increases the ligand’s negative charge and enhances coordination strength toward metal ions. Historically, the development of aminopolycarboxylate chelating agents such as EDTA and CDTA significantly advanced analytical and industrial chemistry by enabling precise control of metal ion concentrations. CDTA became particularly important where stronger or more selective complexation than EDTA was required. Overall, 1,2-cyclohexylenedinitrilotetraacetic acid is a rigid hexadentate aminopolycarboxylate chelating agent with strong metal-binding capability. Its cyclic structure and multiple donor atoms enable formation of highly stable metal complexes, making it valuable in analytical chemistry, coordination chemistry, and metal ion control applications. References 2026. Lithium metal-mediated electrochemical reduction of per- and poly-fluoroalkyl substances. Nature Chemistry. DOI: 10.1038/s41557-025-02057-7 2025. Extraction of Rare Earth Elements and Americium with a Solution of Zirconium Salt of 2-Ethylhexyl Hydrogen 2-Ethylhexylphosphonate in Hydrocarbons and Its Mathematical Description. Radiochemistry. DOI: 10.1134/s1066362225040071 |
| Market Analysis Reports |