Online Database of Chemicals from Around the World
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhejiang Hisun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hisunpharm.com | |||
 | +86 13651899693 | |||
 | yiwen.sun@hisunpharm.com | |||
| Chemical manufacturer since 1956 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Biochemical >> Peptide |
|---|---|
| Name | PF 1022A |
| Synonyms | PF 1022; Cyclo(D-2-hydroxypropanoyl-N-methyl-L-leucyl-3-phenyl-D-2-hydroxypropanoyl-N-methyl-L-leucyl-D-2-hydroxypropanoyl-N-methyl-L-leucyl-3-phenyl-D-2-hydroxypropanoyl-N-methyl-L-leucyl) |
| Molecular Structure | 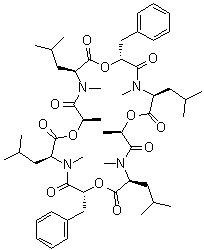 |
| Molecular Formula | C52H76N4O12 |
| Molecular Weight | 949.18 |
| CAS Registry Number | 133413-70-4 |
| EC Number | 444-350-6 |
| SMILES | C[C@@H]1C(=O)N([C@H](C(=O)O[C@@H](C(=O)N([C@H](C(=O)O[C@@H](C(=O)N([C@H](C(=O)O[C@@H](C(=O)N([C@H](C(=O)O1)CC(C)C)C)CC2=CC=CC=C2)CC(C)C)C)C)CC(C)C)C)CC3=CC=CC=C3)CC(C)C)C |
| Solubility | Very slightly soluble (0.91 g/L) (25 °C), Calc.*, CHCL3: 37mg/mL (Expl.) |
|---|---|
| Density | 1.070±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 89-92 °C** |
| Boiling point | 1086.6±65.0 °C 760 mmHg (Calc.)* |
| Flash point | 610.9±34.3 °C (Calc.)* |
| Index of refraction | 1.49 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2017 ACD/Labs) |
| ** | Nakatsu, Shingo |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H319-H413 Details | ||||||||||||
| Safety Statements | P264+P265-P273-P280-P305+P351+P338-P337+P317-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
|
PF 1022A, with the molecular formula C52H76N4O12 and CAS number 133413-70-4, is a cyclooctadepsipeptide isolated from the fungus Mycelia sterilia (FERM BP-2671). It is recognized in medicinal chemistry for its potent anthelmintic properties, targeting parasitic nematodes in veterinary applications. Its discovery and applications are well-documented in the literature, stemming from the exploration of microbial metabolites with antiparasitic activity. The discovery of PF 1022A occurred in the late 1980s by researchers at Meiji Seika Kaisha, Ltd., in Japan, during a screening program for novel anthelmintic compounds produced by microorganisms. The compound was isolated from the fermentation broth of Mycelia sterilia, a fungal strain found in the microflora of the plant Camellia japonica. Its identification addressed the growing need for effective treatments against parasitic nematodes, particularly in livestock and poultry, amid rising resistance to existing anthelmintics like benzimidazoles and avermectins. The unique cyclooctadepsipeptide structure of PF 1022A, distinct from other known anthelmintics, marked it as a promising lead. By 1992, its structure and anthelmintic activity were fully characterized, leading to further research into its derivatives, such as emodepside, a semi-synthetic drug approved for veterinary use. Synthetically, PF 1022A is produced via fermentation of Mycelia sterilia under controlled conditions, typically in a nutrient-rich medium containing glucose, soybean oil, and inorganic salts. The fungus is cultured aerobically, and the compound is extracted from the mycelial biomass using organic solvents like methanol or ethyl acetate, followed by purification through silica gel chromatography and crystallization. The chemical structure, cyclo(D-lactyl-L-N-methylleucyl-D-3-phenyllactyl-L-N-methylleucyl-D-lactyl-L-N-methylleucyl-D-3-phenyllactyl-L-N-methylleucyl), consists of alternating D-lactic acid, D-3-phenyllactic acid, and N-methyl-L-leucine units, forming a 24-membered macrocycle. Chemical synthesis of PF 1022A and its analogs has been achieved using solid-phase peptide synthesis or solution-phase cyclization, such as heating N-methyloctadepsipeptides on oxime resin in refluxing ethyl acetate for two days, though fermentation remains the primary production method due to its efficiency. The primary application of PF 1022A is as an anthelmintic agent in veterinary medicine. It exhibits broad-spectrum activity against gastrointestinal nematodes, including Ascaridia galli in chickens, Haemonchus contortus, Ostertagia ostertagi, and Trichostrongylus colubriformis in ruminants, with effective doses as low as 0.11–0.33 mg/animal in jird models. Its mechanism involves acting as a channel-forming ionophore, disrupting ion transport across nematode cell membranes, and binding to latrophilin-like receptors critical for pharyngeal pumping, leading to paralysis and death of the parasite. PF 1022A also shows activity against the extraintestinal nematode Angiostrongylus cantonensis, though its efficacy against filarial worms like Litomosoides sigmodontis and Brugia malayi is limited, primarily affecting microfilariae. Its derivative, emodepside (Bay 44-4400), developed by Bayer, enhances filaricidal activity and is used in commercial anthelmintic formulations for cats and dogs. PF 1022A is also investigated for angiostrongyliasis research, demonstrating efficacy against Angiostrongylus costaricensis in mice. In academic research, PF 1022A serves as a model for studying cyclodepsipeptide biosynthesis and structure-activity relationships. Its N-methylated leucine residues are critical for high in vivo activity, and modifications have led to analogs with varying anthelmintic potency. The compound’s ionophoric properties and receptor interactions are studied to understand novel anthelmintic mechanisms, addressing resistance to traditional drugs. Its synthesis has advanced techniques in cyclic peptide chemistry, including flow chemistry and solid-phase cyclization. The significance of PF 1022A lies in its introduction of a novel anthelmintic class with a unique mode of action, offering an alternative to conventional treatments. Its development reflects progress in microbial fermentation and peptide chemistry, contributing to veterinary therapeutics and resistance management. References 992. A new anthelmintic cyclodepsipeptide, PF1022A. The Journal of Antibiotics, 45(5). DOI: 10.7164/antibiotics.45.692 1994. Synthesis of PF1022A, an anthelmintic cyclodepsipeptide. The Journal of Antibiotics, 47(11). DOI: 10.7164/antibiotics.47.1322 2000. Biosynthesis of PF1022A and Related Cyclooctadepsipeptides. The Journal of Biological Chemistry, 275(23). DOI: 10.1074/jbc.m001084200 |
| Market Analysis Reports |