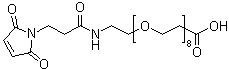
1-Maleimido-3-oxo-7,10,13,16,19,22,25,28-octaoxa-4-aza-hentriacontan-31-oic acid is a complex organic compound characterized by a large polyether backbone, containing both oxygen and nitrogen atoms, along with a maleimido group and a carboxylic acid functional group. The structure consists of a long chain with alternating ether linkages, nitrogen atoms, and functional groups at specific positions along the chain.
This compound belongs to the family of functionalized polyethers and is of interest in a variety of fields, including material science, medicinal chemistry, and nanotechnology. The inclusion of a maleimide group and a carboxyl group at the termini of the molecule enhances its reactivity, making it a versatile building block for further modifications and functionalization.
The maleimido group, characterized by a reactive double bond with the nitrogen, can undergo a variety of chemical reactions, particularly the Michael addition reaction. This allows the compound to act as a coupling agent or crosslinking agent in the synthesis of more complex molecules or polymers. The carboxylic acid group provides another reactive site that can participate in esterification or amidation reactions, facilitating further modifications or conjugations with other molecules, such as drugs or targeting ligands.
In addition to its chemical reactivity, the polyether backbone with nitrogen atoms can enhance the solubility and stability of the compound in aqueous environments, making it potentially useful in biological applications. The ether linkages and the nitrogen atoms in the structure may also contribute to its ability to interact with various biomolecules, such as proteins, nucleic acids, or other macromolecules, which is beneficial for drug delivery systems or biomolecular recognition.
The octaoxa and aza-ether segments of the molecule could further contribute to its ability to form stable complexes with metal ions, enabling its use in the development of metal-organic frameworks (MOFs) or other coordination complexes. These materials have wide-ranging applications in catalysis, sensors, and storage of gases or other substances.
Due to the functional groups present, 1-maleimido-3-oxo-7,10,13,16,19,22,25,28-octaoxa-4-aza-hentriacontan-31-oic acid is also a candidate for use in drug delivery systems. The maleimide group can be employed to conjugate therapeutic molecules to the polyether backbone, facilitating the transport of drugs or other bioactive agents to specific target sites in the body. Additionally, the carboxyl group could be involved in the formation of stable conjugates with targeting molecules or polyethylene glycol (PEG), enhancing the pharmacokinetics and bioavailability of the conjugated drug.
In conclusion, 1-maleimido-3-oxo-7,10,13,16,19,22,25,28-octaoxa-4-aza-hentriacontan-31-oic acid is a functionalized polyether compound with significant potential for use in organic synthesis, nanotechnology, and medicinal chemistry. Its ability to participate in various chemical reactions, its solubility in aqueous environments, and its potential for drug delivery applications make it a versatile molecule in research and industrial applications. The unique combination of functional groups and the large polyether backbone further enhances its value in the development of new materials and therapeutic agents.
|










 GHS07 Warning Details
GHS07 Warning Details